Featured
-
-
Article
| Open Access Structure of a photosynthetic reaction centre determined by serial femtosecond crystallography
Structure of a photosynthetic reaction centre determined by serial femtosecond crystallography
Serial femtosecond crystallography is an X-ray free-electron-laser-based method that uses X-ray bursts to determine protein structures. Here the authors present the structure of a photosynthetic reaction centre, an integral membrane protein, achieved with no sign of X-ray-induced radiation damage.
- Linda C. Johansson
- , David Arnlund
- & Richard Neutze
-
Article
| Open Access Structural basis of AMPK regulation by small molecule activators
Structural basis of AMPK regulation by small molecule activators
Small molecule activators of the energy sensing kinase AMPK are promising candidates as therapies for metabolic disease. Xiao et al. present the crystal structure of AMPK in complex with a small molecule activator, and show that the drug stabilizes interaction between the catalytic and carbohydrate-binding domains.
- Bing Xiao
- , Matthew J. Sanders
- & Steven J. Gamblin
-
Article
| Open Access Structural and molecular basis of ZNRF3/RNF43 transmembrane ubiquitin ligase inhibition by the Wnt agonist R-spondin
Structural and molecular basis of ZNRF3/RNF43 transmembrane ubiquitin ligase inhibition by the Wnt agonist R-spondin
R-spondins are secreted factors that potentiate Wnt signalling by inhibiting the degradation of Wnt receptors by transmembrane E3 ubiquitin ligases. Zebisch et al.present a panel of crystal structures that reveal how this inhibition occurs.
- Matthias Zebisch
- , Yang Xu
- & E. Yvonne Jones
-
Article |
 Structural insight into the mutual recognition and regulation between Suppressor of Fused and Gli/Ci
Structural insight into the mutual recognition and regulation between Suppressor of Fused and Gli/Ci
Suppressor of Fused (Sufu) controls the nucleocytoplasmic distribution of Gli transcription factors in response to Hedgehog signalling. Zhang et al.report crystal structures of the Sufu/Gli complex, and show that Sufu transitions from an open to a closed form in response to Gli binding.
- Yan Zhang
- , Lin Fu
- & Geng Wu
-
Article |
 Structural insight into dGTP-dependent activation of tetrameric SAMHD1 deoxynucleoside triphosphate triphosphohydrolase
Structural insight into dGTP-dependent activation of tetrameric SAMHD1 deoxynucleoside triphosphate triphosphohydrolase
SAMHD1 is a dNTP hydrolase that has been shown to act as a restriction factor against retroviruses such as HIV, and also regulates the retrotransposition of LINE-1 elements. Here the authors reveal the structural basis for dGTP-dependent tetramerization and allosteric activation of the enzyme.
- Chunfeng Zhu
- , Wenying Gao
- & Xiao-Fang Yu
-
Article |
 Structural basis of allosteric interactions among Ca2+-binding sites in a K+ channel RCK domain
Structural basis of allosteric interactions among Ca2+-binding sites in a K+ channel RCK domain
Regulator of K+ conductance (RCK) domains control the gating of potassium channels in response to ligands such as calcium. Smith et al. solve structures of the MthK RCK domain with varying numbers of calcium ions bound, and reveal the structural basis of allosteric coupling between calcium binding sites.
- Frank J. Smith
- , Victor P.T. Pau
- & Brad S. Rothberg
-
Article |
 Structure of GrlR–GrlA complex that prevents GrlA activation of virulence genes
Structure of GrlR–GrlA complex that prevents GrlA activation of virulence genes
The locus of enterocyte effacement is a pathogenicity island essential for virulence of some Escherichia colipathogenic serotypes. Here, the authors solve the structure of a regulatory complex that optimizes regulation of this locus during the infection process.
- Abhilash Padavannil
- , Chacko Jobichen
- & J. Sivaraman
-
Article |
 CARMIL leading edge localization depends on a non-canonical PH domain and dimerization
CARMIL leading edge localization depends on a non-canonical PH domain and dimerization
CARMIL regulates actin capping proteins at the leading edge of migrating cells. By solving the crystal structure of the CARMIL N-terminal region, Zwolak et al. discover that this region encodes a cryptic pleckstrin homology domain, and show that it mediates association with the plasma membrane.
- Adam Zwolak
- , Changsong Yang
- & Roberto Dominguez
-
Article
| Open Access Role of the C-terminal domain in the structure and function of tetrameric sodium channels
Role of the C-terminal domain in the structure and function of tetrameric sodium channels
Prokaryotic voltage-gated sodium channels possess dynamically disordered cytoplasmic C-terminal domains. Bagnéris et al. present the structure of the Magnetococcus marinusNavMs pore and C-terminal domain and reveal its role in coupling channel inactivation and opening.
- Claire Bagnéris
- , Paul G. DeCaen
- & B. A. Wallace
-
Article
| Open Access Visualization of poly(ADP-ribose) bound to PARG reveals inherent balance between exo- and endo-glycohydrolase activities
Visualization of poly(ADP-ribose) bound to PARG reveals inherent balance between exo- and endo-glycohydrolase activities
Poly-ADP-ribosylation is a post-translational modification that is countered by poly(ADP-ribose) glycohydrolases (PARGs). In this study, the authors present the crystal structure of poly(ADP-ribose) glycohydrolase (PARGs) in complex with a poly(ADP-ribose) substrate, and reveal that poly(ADP-ribose) glycohydrolase (PARGs) enzymes act predominantly as exo- rather than as endo-glycohydrolases.
- Eva Barkauskaite
- , Amy Brassington
- & David Leys
-
Article |
 Tertiary structural elements determine the extent and specificity of messenger RNA editing
Tertiary structural elements determine the extent and specificity of messenger RNA editing
A central, imperfect duplex RNA secondary structure is generally required for site-specific adenosine-to-inosine RNA editing by ADAR enzymes. Rieder et al. show in Drosophila that conserved and complex long-range RNA tertiary structures form in vivoand can also regulate specific RNA-editing events by ADAR enzymes.
- Leila E. Rieder
- , Cynthia J. Staber
- & Robert A. Reenan
-
Article |
 Crystal structure of the yeast TSC1 core domain and implications for tuberous sclerosis pathological mutations
Crystal structure of the yeast TSC1 core domain and implications for tuberous sclerosis pathological mutations
Tuberous sclerosis is a disease characterized by tumour-like growths in multiple organs and is caused by mutations in TSC1 or TSC2. Here, the authors solve the crystal structure of yeast TSC1 and find that most mutations are found inside the folded N-terminal domain.
- Wei Sun
- , Ye Julia Zhu
- & Wenqing Xu
-
Article |
 Recognition of vitamin B metabolites by mucosal-associated invariant T cells
Recognition of vitamin B metabolites by mucosal-associated invariant T cells
T-cell receptors expressed on mucosal-associated invariant T cells function in a similar manner to innate immune receptors by recognizing small molecules such as microbial metabolites. Here, the authors report structures of this receptor in complex with vitamin B metabolites presented by the MHC-like protein MR1.
- Onisha Patel
- , Lars Kjer-Nielsen
- & Jamie Rossjohn
-
Article |
 Structure of dual receptor binding to botulinum neurotoxin B
Structure of dual receptor binding to botulinum neurotoxin B
Botulinum neurotoxin B binds to two neuronal surface receptors, synaptotagmin II and ganglioside receptor GD1a. Berntsson et al.present the crystal structure of botulinum neurotoxin B in complex with both receptor proteins, demonstrating independent binding.
- Ronnie P-A. Berntsson
- , Lisheng Peng
- & Pål Stenmark
-
Article
| Open Access The crystal structure of multidrug-resistance regulator RamR with multiple drugs
The crystal structure of multidrug-resistance regulator RamR with multiple drugs
RamR is an important multidrug-resistance factor, however, its structure and the molecules to which it responds are hitherto unknown. Here, the authors report the crystal structures of RamR complexed with multiple drugs, revealing significant flexibility in its substrate-recognition region.
- Suguru Yamasaki
- , Eiji Nikaido
- & Kunihiko Nishino
-
Article |
 Antagonism between binding site affinity and conformational dynamics tunes alternative cis-interactions within Shp2
Antagonism between binding site affinity and conformational dynamics tunes alternative cis-interactions within Shp2
Competition between inter- and intra-molecular interactions is a commonly observed property of modular signalling proteins. Here, the authors show that in the phosphatase Shp2, this balance is maintained by a trade-off between binding site affinities and conformational stability.
- Jie Sun
- , Shaoying Lu
- & Yingxiao Wang
-
Article
| Open Access A molecular explanation for the recessive nature of parkin-linked Parkinson’s disease
A molecular explanation for the recessive nature of parkin-linked Parkinson’s disease
Mutations in the E3 ubiquitin ligase parkin are associated with juvenile Parkinson’s disease. Here the authors report the solution structure of the Parkin RING2 domain, revealing how disease-associated mutations affect its function and providing a molecular explanation for the recessive nature of the disease.
- Donald E. Spratt
- , R Julio Martinez-Torres
- & Gary S. Shaw
-
Article
| Open Access Structure and function of Parkin E3 ubiquitin ligase reveals aspects of RING and HECT ligases
Structure and function of Parkin E3 ubiquitin ligase reveals aspects of RING and HECT ligases
The Parkinson’s disease-associated protein Parkin regulates the fate of damaged mitochondria by ubiquitinating mitochondrial substrates. Riley et al. present the crystal structure of the Parkin-R0RBR domain, providing new insight into the catalytic mechanism of the enzyme.
- B.E. Riley
- , J.C. Lougheed
- & J.A. Johnston
-
Article |
 NMR structure of human restriction factor APOBEC3A reveals substrate binding and enzyme specificity
NMR structure of human restriction factor APOBEC3A reveals substrate binding and enzyme specificity
The cytidine deaminase APOBEC3A has potent antiviral activity, degrading foreign DNA, and inhibiting viral replication, retrotransposition and reverse transcription. Byeon et al. present the solution structure of APOBEC3A, and reveal insights into its substrate specificity.
- In-Ja L. Byeon
- , Jinwoo Ahn
- & Angela M. Gronenborn
-
Article |
 Crystal structures of interleukin 17A and its complex with IL-17 receptor A
Crystal structures of interleukin 17A and its complex with IL-17 receptor A
Interleukin-17A homodimers preferentially interact with heterodimeric IL-17 receptors. By solving the crystal structure of an IL-17A homodimer in complex with a single IL-17RA receptor subunit, the authors reveal conformational changes in IL-17A that lead to exclusion of a second IL-17RA subunit.
- Shenping Liu
- , Xi Song
- & Matthew C. Griffor
-
Article
| Open Access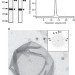 The four-transmembrane protein IP39 of Euglena forms strands by a trimeric unit repeat
The four-transmembrane protein IP39 of Euglena forms strands by a trimeric unit repeat
IP39 is an abundant protozoan protein known to form highly-ordered striations in Euglena gracilis’ plasma membrane. Here, Suzuki et al. determine its three-dimensional structure by electron crystallography revealing that IP39 polymerises to form trimeric longitudinal units arranged in a molecular strand of antiparallel double-rows.
- Hiroshi Suzuki
- , Yasuyuki Ito
- & Sachiko Tsukita
-
Article |
 Structural basis for potentiation by alcohols and anaesthetics in a ligand-gated ion channel
Structural basis for potentiation by alcohols and anaesthetics in a ligand-gated ion channel
Alcohols and anaesthetics exert their effects by potentiating ligand-gated ion channels. Here, the authors determine crystal structures of a bacterial ligand-gated ion channel in the presence of alcohols and anaesthetics, and describe a structural mechanism for stabilization of the open form of the channel.
- Ludovic Sauguet
- , Rebecca J. Howard
- & Marc Delarue
-
Article
| Open Access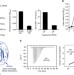 Structural basis of protein phosphatase 2A stable latency
Structural basis of protein phosphatase 2A stable latency
The protein α4 is essential for the formation, stability and activity of protein phosphatase 2A (PP2A) complexes. Here the authors solve the crystal structure of a truncated PP2A bound to α4 and show that α4 binds to a partially folded form of the protein, stabilizing the enzyme in an inactive state.
- Li Jiang
- , Vitali Stanevich
- & Yongna Xing
-
Article |
 Energetics of activation of GTP hydrolysis on the ribosome
Energetics of activation of GTP hydrolysis on the ribosome
Ribosomal protein synthesis is driven by the hydrolysis of GTP. Wallin and colleagues employ molecular dynamics and computer simulations to show that a universally conserved histidine promotes GTP hydrolysis in its protonated form, and is driven into the active conformation by interactions with the ribosome.
- Göran Wallin
- , Shina C. L. Kamerlin
- & Johan Åqvist
-
Article |
 Structure of ERK2 bound to PEA-15 reveals a mechanism for rapid release of activated MAPK
Structure of ERK2 bound to PEA-15 reveals a mechanism for rapid release of activated MAPK
PEA-15 is a scaffold protein that regulates the localization and phosphorylation of the MAP kinase ERK2. By solving the structure of the PEA-15/ERK2 complex, the authors show that PEA-15 restrains ERK2 in a spring-loaded, activated form.
- Peter D. Mace
- , Yann Wallez
- & Stefan J. Riedl
-
Article |
 Two-dimensional structure from random multiparticle X-ray scattering images using cross-correlations
Two-dimensional structure from random multiparticle X-ray scattering images using cross-correlations
A model-independent protocol is developed for reconstructing a two-dimensional particle from multiparticle diffraction images. The method is verified experimentally with gold nanostructures, paving the way towards single-molecule imaging using high-intensity X-ray free-electron laser pulses.
- B. Pedrini
- , A. Menzel
- & R. Abela
-
Article |
 Structural basis for recognition of autophagic receptor NDP52 by the sugar receptor galectin-8
Structural basis for recognition of autophagic receptor NDP52 by the sugar receptor galectin-8
When Salmonella invades human cells, galectin-8 binds host glycans exposed on damaged vesicles and recruits NDP52 to trigger autophagy of the invading microbe. Kim et al. reveal the structure of the NDP52–galectin-8 complex and the basis for its carbohydrate specificity.
- Byeong-Won Kim
- , Seung Beom Hong
- & Hyun Kyu Song
-
Article |
 Structure of the full-length HCV IRES in solution
Structure of the full-length HCV IRES in solution
The hepatitis C virus RNA genome is translated via an internal ribosome entry site. Pérard et al. present an atomic model of this site, and using molecular dynamics simulations, identify conformational flexibility that may underlie its function during translation initiation.
- Julien Pérard
- , Cédric Leyrat
- & Marc Jamin
-
Article |
 Crystal structure of human tyrosylprotein sulfotransferase-2 reveals the mechanism of protein tyrosine sulfation reaction
Crystal structure of human tyrosylprotein sulfotransferase-2 reveals the mechanism of protein tyrosine sulfation reaction
The post-translational protein modification tyrosine sulfation is catalysed by tyrosylprotein sulfotransferase (TPST). Teramoto et al. present the first crystal structure of the human TPST isoform 2 complexed with a substrate peptide derived from complement C4 and 3′phosphoadenosine-5′-phosphate, revealing the molecular basis of catalysis.
- Takamasa Teramoto
- , Yukari Fujikawa
- & Yoshimitsu Kakuta
-
Article |
 Mechanism of tetracycline resistance by ribosomal protection protein Tet(O)
Mechanism of tetracycline resistance by ribosomal protection protein Tet(O)
The bacterial tetracycline resistance protein Tet(O) binds to the ribosome, preventing tetracycline from inhibiting translation. Using cryo-electron microscopic reconstruction, the authors present an atomic model of Tet(O) bound to the 70S ribosome, and reveal how Tet(O) promotes antibiotic resistance.
- Wen Li
- , Gemma C. Atkinson
- & Joachim Frank
-
Article |
 Self-organization of the vesicular stomatitis virus nucleocapsid into a bullet shape
Self-organization of the vesicular stomatitis virus nucleocapsid into a bullet shape
The bullet shape of Rhabdoviruses is thought to depend on their matrix protein, which stabilizes the nucleocapsid coil. Desfosses and colleagues show that the nucleoprotein of the vesicular stomatitis virus can fold the genome into a bullet-shaped skeleton in the absence of other viral components.
- Ambroise Desfosses
- , Euripedes A. Ribeiro Jr
- & Irina Gutsche
-
Article
| Open Access Structural insights into protein-only RNase P complexed with tRNA
Structural insights into protein-only RNase P complexed with tRNA
RNase P is a key enzyme implicated in transfer RNA maturation that removes the 5′-leader sequences from transfer RNA precursors. In this study, a biophysical characterization of a novel protein-only variant of RNase P, known as PRORP (PROteinaceous RNase P), reveals that transfer RNA recognition by PRORP is similar to that by ribonucleoprotein RNase P.
- Anthony Gobert
- , Franziska Pinker
- & Philippe Giegé
-
Article |
 A structural basis for streptomycin-induced misreading of the genetic code
A structural basis for streptomycin-induced misreading of the genetic code
The antibiotic streptomycin increases errors in protein translation, but it is unclear how streptomycin exerts its effect on the ribosome. Demirci et al. present X-ray crystal structures that reveal conformational changes induced by streptomycin, which may inspire future efforts in antibiotics design.
- Hasan Demirci
- , Frank Murphy IV
- & Gerwald Jogl
-
Article
| Open Access Alba shapes the archaeal genome using a delicate balance of bridging and stiffening the DNA
Alba shapes the archaeal genome using a delicate balance of bridging and stiffening the DNA
How the genome is physically organized is less understood in archaea than in eubacteria or eukaryotes. Laurens et al. measure DNA binding by the Sulfolobus solfataricusproteins Alba1 and Alba2 using single-molecule techniques and conclude that the presence of Alba2 leads to more bridging between DNA.
- Niels Laurens
- , Rosalie P.C. Driessen
- & Gijs J.L. Wuite
-
Article |
 Catalytic site remodelling of the DOT1L methyltransferase by selective inhibitors
Catalytic site remodelling of the DOT1L methyltransferase by selective inhibitors
Selective inhibitors of protein methyltransferases are anticancer drug candidates. Yu et al. report the structural changes that occur when selective inhibitors bind to the protein methyltransferase DOT1L.
- Wenyu Yu
- , Emma J. Chory
- & Matthieu Schapira
-
Article
| Open Access MPIase is a glycolipozyme essential for membrane protein integration
MPIase is a glycolipozyme essential for membrane protein integration
Proteins are integrated into cellular membranes either co-translationally through Sec/SRP or post-translationally by chaperones. These authors show that an integration-dedicated chaperone inE. coli, MPIase, is a glycolipid and facilitates protein insertion into the inner membrane of the bacterium.
- Ken-ichi Nishiyama
- , Masahide Maeda
- & Keiko Shimamoto
-
Article
| Open Access Structural and molecular insights into the mechanism of action of human angiogenin-ALS variants in neurons
Structural and molecular insights into the mechanism of action of human angiogenin-ALS variants in neurons
Mutations in human angiogenin are implicated in the progression of amyotrophic lateral sclerosis. Thiyagarajan and colleagues show that structural differences between angiogenin variants affect neuronal survival, and the ability to induce stress granules in neuronal cell lines.
- Nethaji Thiyagarajan
- , Ross Ferguson
- & K. Ravi Acharya
-
Article
| Open Access Structure of a bacterial voltage-gated sodium channel pore reveals mechanisms of opening and closing
Structure of a bacterial voltage-gated sodium channel pore reveals mechanisms of opening and closing
Sodium-gated ion channels open and close in response to the flow of ions. Here, McCusker et al.report the open structure of a sodium-gated ion channel pore from a bacterial homologue, and show, by comparison with the closed structure, that the movement of a C-terminal helix is sufficient to open the channel.
- Emily C. McCusker
- , Claire Bagnéris
- & B.A. Wallace
-
Article |
 Discovery of acetylene hydratase activity of the iron–sulphur protein IspH
Discovery of acetylene hydratase activity of the iron–sulphur protein IspH
The iron–sulphur enzyme IspH catalyses the final step of the methylerythritol phosphate isoprenoid biosynthesis pathway. Spanet al. report that IspH can hydrate acetylenes to aldehydes and ketones, in addition to its role as a 2H+/2e−reductase.
- Ingrid Span
- , Ke Wang
- & Michael Groll
-
Article |
 Structures of Pup ligase PafA and depupylase Dop from the prokaryotic ubiquitin-like modification pathway
Structures of Pup ligase PafA and depupylase Dop from the prokaryotic ubiquitin-like modification pathway
Pupylation is a bacterial posttranslational modification pathway with functional analogies to ubiquitination. Here, Özceliket al.report the structures of the Pup Ligase, PafA and the Depupylase, Dop. Mutational analysis revealed residues required for catalysis and for the interaction with Pup.
- Dennis Özcelik
- , Jonas Barandun
- & Eilika Weber-Ban
-
Article |
 Mechanism of resilin elasticity
Mechanism of resilin elasticity
Resilin is a polymeric elastic protein that is important for the flight and jumping of insects. Here, the structure-function relationships ofDrosophilaresilin are investigated, and a mechanical model is proposed to account for its elasticity.
- Guokui Qin
- , Xiao Hu
- & David L. Kaplan
-
Article |
 Comprehensive interrogation of natural TALE DNA-binding modules and transcriptional repressor domains
Comprehensive interrogation of natural TALE DNA-binding modules and transcriptional repressor domains
The peptide sequence of transcription activator-like effectors (TALEs) can be customized to tailor the binding of TALEs to specific DNA sequences. Conget al. improve TALE specificity for guanine binding and use a genetic construct based on TALEs to efficiently repress expression of a target gene.
- Le Cong
- , Ruhong Zhou
- & Feng Zhang
-
Article |
 The yeast regulator of transcription protein Rtr1 lacks an active site and phosphatase activity
The yeast regulator of transcription protein Rtr1 lacks an active site and phosphatase activity
Regulator of transcription 1, Rtr1, has been proposed to function as an RNA polymerase II phosphatase. Here, the lack of phosphatase activity in purified Rtr1 and the absence of an active site in the structure of the yeast protein suggest that Rtr1 does not dephosphorylate RNA polymerase II.
- Kehui Xiang
- , James L. Manley
- & Liang Tong
-
Article
| Open Access Structure of the Acid-sensing ion channel 1 in complex with the gating modifier Psalmotoxin 1
Structure of the Acid-sensing ion channel 1 in complex with the gating modifier Psalmotoxin 1
The activity of the Acid sensing ion channel 1, ASIC1, can be modulated by the gating modifier Psalmotoxin 1 but the molecular mechanism is unclear. Dawsonet al. report the structure of chicken ASIC1 bound to Psalmotoxin 1 and find that the toxin locks two regulatory regions of the channel into a desensitized conformation.
- Roger J.P. Dawson
- , Jörg Benz
- & Armin Ruf
-
Article |
 Crystal structure of a plectonemic RNA supercoil
Crystal structure of a plectonemic RNA supercoil
Nucleic acid superstructures are required to package genomes into the nucleus of cells. In this study, the superstructure of an RNA supercoil species is reported and is shown to be dependent on an RNA-binding protein that induces a higher level of organization compared with DNA superstructures.
- Jason R. Stagno
- , Buyong Ma
- & Xinhua Ji
-
Article |
 Structure and mechanism of a canonical poly(ADP-ribose) glycohydrolase
Structure and mechanism of a canonical poly(ADP-ribose) glycohydrolase
Poly(ADP-ribose) glycohydrolase catabolises poly(ADP-ribose), which is covalently attached to proteins following post-translational modification. In this study, the structure of poly(ADP-ribose) glycohydrolase fromTetrahymena thermophilais reported in complex with the small molecule inhibitor RBPI-3.
- Mark S. Dunstan
- , Eva Barkauskaite
- & Ivan Ahel
-
Article |
 Assembly kinetics determine the architecture of α-actinin crosslinked F-actin networks
Assembly kinetics determine the architecture of α-actinin crosslinked F-actin networks
Processes that regulate the mechanical and dynamic properties of actin cytoskeleton are important for multiple eukaryotic cell functions. In this study, using reconstituted crosslinked actin networks, organization is found to be determined by the competing kinetics of actin gelation and bundle formation.
- Tobias T. Falzone
- , Martin Lenz
- & Margaret L. Gardel
-
Article
| Open Access The cytoplasmic coiled-coil mediates cooperative gating temperature sensitivity in the voltage-gated H+ channel Hv1
The cytoplasmic coiled-coil mediates cooperative gating temperature sensitivity in the voltage-gated H+ channel Hv1
In the dimeric voltage-gated H+channel, Hv1/VSOP, the gating of each subunit is coupled, but the molecular basis of dimer formation and intersubunit coupling is unclear. This study shows that channel assembly and cooperative gating are mediated by the cytoplasmic domain.
- Yuichiro Fujiwara
- , Tatsuki Kurokawa
- & Yasushi Okamura
-
Article
| Open Access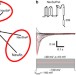 The C-terminal helical bundle of the tetrameric prokaryotic sodium channel accelerates the inactivation rate
The C-terminal helical bundle of the tetrameric prokaryotic sodium channel accelerates the inactivation rate
Many channels have cytosolic domains which regulate channel function. Irieet al. show that the cytosolic C-terminal region of NavSulP, a prokaryotic voltage-gated sodium channel, forms a four-helix bundle which stabilises the tetrameric channel and accelerates channel inactivation.
- Katsumasa Irie
- , Takushi Shimomura
- & Yoshinori Fujiyoshi

 Structural insights into the role of the Smoothened cysteine-rich domain in Hedgehog signalling
Structural insights into the role of the Smoothened cysteine-rich domain in Hedgehog signalling