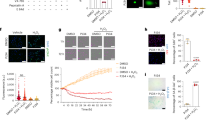
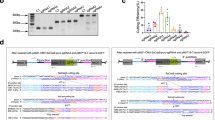
Abstract
Age-related macular degeneration (AMD), a leading cause of blindness, initiates in the outer-blood-retina-barrier (oBRB) formed by the retinal pigment epithelium (RPE), Bruch’s membrane, and choriocapillaris. The mechanisms of AMD initiation and progression remain poorly understood owing to the lack of physiologically relevant human oBRB models. To this end, we engineered a native-like three-dimensional (3D) oBRB tissue (3D-oBRB) by bioprinting endothelial cells, pericytes, and fibroblasts on the basal side of a biodegradable scaffold and establishing an RPE monolayer on top. In this 3D-oBRB model, a fully-polarized RPE monolayer provides barrier resistance, induces choriocapillaris fenestration, and supports the formation of Bruch’s-membrane-like structure by inducing changes in gene expression in cells of the choroid. Complement activation in the 3D-oBRB triggers dry AMD phenotypes (including subRPE lipid-rich deposits called drusen and choriocapillaris degeneration), and HIF-α stabilization or STAT3 overactivation induce choriocapillaris neovascularization and type-I wet AMD phenotype. The 3D-oBRB provides a physiologically relevant model to studying RPE–choriocapillaris interactions under healthy and diseased conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The scRNA-seq data generated in this study have been deposited in the Gene Expression Omnibus database under accession code GSE214928. These RNA-seq data are openly available without any restriction. All the processed data are available within the article. All the raw data generated in this study are provided in Source Data files. Source data for Fig. 6 are available in a figshare repository at https://doi.org/10.6084/m9.figshare.21300198. Source data are provided with this paper.
Code availability
Angiogenesis quantification was performed on MATLAB version 2019b (Mathworks). The custom MATLAB code is available in Supplementary Information.
References
Wong, W. L. et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob. Health 2, e106–e116 (2014).
Song, M. J. & Bharti, K. Looking into the future: using induced pluripotent stem cells to build two and three dimensional ocular tissue for cell therapy and disease modeling. Brain Res. 1638, 2–14 (2016).
McLeod, D. S. et al. Relationship between RPE and choriocapillaris in age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 50, 4982–4991 (2009).
Bhutto, I. & Lutty, G. Understanding age-related macular degeneration (AMD): relationships between the photoreceptor/retinal pigment epithelium/Bruch’s membrane/choriocapillaris complex. Mol. Asp. Med 33, 295–317 (2012).
Farecki, M. L. et al. Characteristics of type 1 and 2 CNV in exudative AMD in OCT-angiography. Graefes Arch. Clin. Exp. Ophthalmol. 255, 913–921 (2017).
Kovach, J. L., Schwartz, S. G., Flynn, H. W. Jr. & Scott, I. U. Anti-VEGF treatment strategies for wet AMD. J. Ophthalmol. 2012, 786870 (2012).
Barben, M., Samardzija, M. & Grimm, C. The role of hypoxia, hypoxia-inducible factor (HIF), and VEGF in retinal angiomatous proliferation. Adv. Exp. Med Biol. 1074, 177–183 (2018).
Manian, K. V. et al. 3D iPSC modeling of the retinal pigment epithelium–choriocapillaris complex identifies factors involved in the pathology of macular degeneration. Cell Stem Cell 28, 978 (2021).
Benedicto, I. et al. Concerted regulation of retinal pigment epithelium basement membrane and barrier function by angiocrine factors. Nat. Commun. 8, 15374 (2017).
Chirco, K. R., Sohn, E. H., Stone, E. M., Tucker, B. A. & Mullins, R. F. Structural and molecular changes in the aging choroid: implications for age-related macular degeneration. Eye (Lond.) 31, 10–25 (2017).
Sweeney, M. & Foldes, G. It takes two: endothelial-perivascular cell cross-talk in vascular development and disease. Front Cardiovasc Med 5, 154 (2018).
Campbell, M. & Humphries, P. The blood-retina barrier: tight junctions and barrier modulation. Adv. Exp. Med Biol. 763, 70–84 (2012).
Nickla, D. L. & Wallman, J. The multifunctional choroid. Prog. Retin Eye Res 29, 144–168 (2010).
Hoshino, A., Chiba, H., Nagai, K., Ishii, G. & Ochiai, A. Human vascular adventitial fibroblasts contain mesenchymal stem/progenitor cells. Biochem. Biophys. Res. Commun. 368, 305–310 (2008).
Orlova, V. V. et al. Generation, expansion and functional analysis of endothelial cells and pericytes derived from human pluripotent stem cells. Nat. Protoc. 9, 1514–1531 (2014).
Sharma, R. et al. Clinical-grade stem cell-derived retinal pigment epithelium patch rescues retinal degeneration in rodents and pigs Sci. Transl. Med. 11, eaat5580 (2019).
Shiihara, H. et al. Quantitative analyses of diameter and running pattern of choroidal vessels in central serous chorioretinopathy by en face images. Sci. Rep. 10, 9591 (2020).
Royston, D. Preventing the inflammatory response to open-heart surgery: the role of aprotinin and other protease inhibitors. Int. J. Cardiol. 53, S11–S37 (1996).
Baltazar, T. et al. Three dimensional bioprinting of a vascularized and perfusable skin graft using human keratinocytes, fibroblasts, pericytes, and endothelial cells. Tissue Eng. Part A 26, 227–238 (2020).
Guillotin, B. et al. Laser assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials 31, 7250–7256 (2010).
Abdeen, A. A., Lee, J., Mo, S. H. & Kilian, K. A. Spatially defined stem cell-laden hydrogel islands for directing endothelial tubulogenesis. J. Mater. Chem. B 3, 7896–7898 (2015).
Lamalice, L., Le Boeuf, F. & Huot, J. Endothelial cell migration during angiogenesis. Circ. Res. 100, 782–794 (2007).
Brindle, N. P., Saharinen, P. & Alitalo, K. Signaling and functions of angiopoietin-1 in vascular protection. Circ. Res. 98, 1014–1023 (2006).
Newman, A. C., Nakatsu, M. N., Chou, W., Gershon, P. D. & Hughes, C. C. The requirement for fibroblasts in angiogenesis: fibroblast-derived matrix proteins are essential for endothelial cell lumen formation. Mol. Biol. Cell 22, 3791–3800 (2011).
Payne, L. B. et al. The pericyte microenvironment during vascular development. Microcirculation 26, e12554 (2019).
Maminishkis, A. et al. Confluent monolayers of cultured human fetal retinal pigment epithelium exhibit morphology and physiology of native tissue. Invest. Ophthalmol. Vis. Sci. 47, 3612–3624 (2006).
Campochiaro, P. A., Jerdon, J. A. & Glaser, B. M. The extracellular matrix of human retinal pigment epithelial cells in vivo and its synthesis in vitro. Invest. Ophthalmol. Vis. Sci. 27, 1615–1621 (1986).
Choi, W. et al. Choriocapillaris and choroidal microvasculature imaging with ultrahigh speed OCT angiography. PLoS One 8, e81499 (2013).
Grebe, R. et al. Ultrastructural analysis of submacular choriocapillaris and its transport systems in AMD and aged control eyes. Exp. Eye Res 181, 252–262 (2019).
Takei, Y. & Ozanics, V. Origin and development of Bruch’s membrane in monkey fetuses: an electron microscopic study. Invest Ophthalmol. 14, 903–916 (1975).
Voigt, A. P. et al. Choroidal endothelial and macrophage gene expression in atrophic and neovascular macular degeneration. Hum. Mol. Genet 31, 2406–2423 (2022).
Kosyakova, N. et al. Differential functional roles of fibroblasts and pericytes in the formation of tissue-engineered microvascular networks in vitro. NPJ Regen. Med 5, 1 (2020).
Curcio, C. J. in Retina Vol. 1 (eds Ryan, S. J.) Ch. 20 (Elsevier, 2013).
Johnson, L. V. et al. Cell culture model that mimics drusen formation and triggers complement activation associated with age-related macular degeneration. Proc. Natl Acad. Sci. USA 108, 18277–18282 (2011).
Galloway, C. A. et al. Drusen in patient-derived hiPSC-RPE models of macular dystrophies. Proc. Natl Acad. Sci. USA 114, E8214–E8223 (2017).
Sharma, R. et al. Epithelial phenotype restoring drugs suppress macular degeneration phenotypes in an iPSC model. Nat. Commun. 12, 7293 (2021).
Vadlapatla, R. K., Vadlapudi, A. D. & Mitra, A. K. Hypoxia-inducible factor-1 (HIF-1): a potential target for intervention in ocular neovascular diseases. Curr. Drug Targets 14, 919–935 (2013).
Mammadzada, P., Corredoira, P. M. & Andre, H. The role of hypoxia-inducible factors in neovascular age-related macular degeneration: a gene therapy perspective. Cell. Mol. Life Sci. 77, 819–833 (2020).
Theriault, J. R. et al. Discovery of a new molecular probe ML228: an activator of the hypoxia inducible factor (HIF) pathway. Bioorg. Med. Chem. Lett. 22, 76–81 (2012).
Yang, J. et al. Two-year risk of exudation in eyes with nonexudative age-related macular degeneration and subclinical neovascularization detected with swept source optical coherence tomography angiography. Am. J. Ophthalmol. 208, 1–11 (2019).
Li, X. et al. Hyperglycaemia exacerbates choroidal neovascularisation in mice via the oxidative stress-induced activation of STAT3 signalling in RPE cells. PLoS ONE 7, e47600 (2012).
Jing Wang, Q. B., et al. Chrysin alleviates DNA damage to improve disturbed immuno-homeostasis and pro-angiogenic environment in laser-induced choroidal neovascularization. Preprint available at Research Square https://doi.org/10.21203/rs.3.rs-827729/v1 (2021).
Wavre-Shapton, S. T., Tolmachova, T., Lopes da Silva, M., Futter, C. E. & Seabra, M. C. Conditional ablation of the choroideremia gene causes age-related changes in mouse retinal pigment epithelium. PLoS One 8, e57769 (2013).
Booij, J. C., Baas, D. C., Beisekeeva, J., Gorgels, T. G. & Bergen, A. A. The dynamic nature of Bruch’s membrane. Prog. Retin. Eye Res. 29, 1–18 (2010).
Huang, J. D., Presley, J. B., Chimento, M. F., Curcio, C. A. & Johnson, M. Age-related changes in human macular Bruch’s membrane as seen by quick-freeze/deep-etch. Exp. Eye Res. 85, 202–218 (2007).
Farazdaghi, M. K. & Ebrahimi, K. B. Role of the choroid in age-related macular degeneration: a current review. J. Ophthalmic Vis. Res 14, 78–87 (2019).
Cavallotti, C., Artico, M., Pescosolido, N., Leali, F. M. & Feher, J. Age-related changes in the human retina. Can. J. Ophthalmol. 39, 61–68 (2004).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 e1821 (2019).
Zhou, W. et al. Single-cell analysis reveals regulatory gene expression dynamics leading to lineage commitment in early T cell development. Cell Syst. 9, 321–337 (2019).
Chen, E. Y. et al. Enrichr: interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinf. 14, 128 (2013).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90–W97 (2016).
Xie, Z. et al. Gene set knowledge discovery with Enrichr. Curr. Protoc. 1, e90 (2021).
Cowan, C. S. et al. Cell types of the human retina and its organoids at single-cell resolution. Cell 182, 1623–1640 (2020).
Song, H. W. et al. Transcriptomic comparison of human and mouse brain microvessels. Sci. Rep. 10, 12358 (2020).
McGinnis, C. S., Murrow, L. M. & Gartner, Z. J. DoubletFinder: doublet detection in single-cell RNA sequencing data using artificial nearest neighbors. Cell Syst. 8, 329–337 (2019).
Safran, M. et al. in Practical Guide to Life Science Databases (eds Abugessaisa, I. & Kasukawa, T.) 27–56 (Springer Nature, 2021).
Acknowledgements
The authors thank R. Fariss, NEI Biological Imaging Core, M. Abu from NEI histology core for processing the samples for TEM and taking images, D. McGaughey OGVFB, NEI for advice on scRNA-seq data analysis. This work was supported by funds from the NEI Intramural Research Program to K.B., NCATS intramural funds to M.J.S., M.F., and I.S., NHLBI intramural funds to M.B., Department of Defense grant (grant number 11831456) to M.J.S. and K.B., and Cures Acceleration Network program to M.J.S. and M.F.
Author information
Authors and Affiliations
Contributions
Conceptualization: K.B., M.J.S, and M.F.; methodology: K.B., R.Q., E.N, M.J.S, T.V., I.S., and M.F.; Investigation: R.Q., E.N, M.J.S, C.H., R.S., T.S.P., C.K., C.T., C.W., A.S., R.D., D.B., P.D., K.D., S.M., G. C., M.B., A.M., and F.B.; analysis: Y.-C.C., K.B., R.Q., E.N, and M.J.S.; project administration: R.Q., E.N, and M.J.S; writing, review, and editing: K.B., E.N, M.J.S, and M.F; funding acquisition: K.B. M.F., M.B., I.S., and M.J.S.; resources: K.B. and M.F.; supervision: K.B. and M.F.
Corresponding author
Ethics declarations
Competing interests
The authors have an approved patent on this technology in Australia (#AU2017359330B2) and a pending patent in the US (#US20190290803A1).
Peer review
Peer review information
Nature Methods thanks Reinhold Medina, Botond Roska, Ruchira Singh for their contribution to the peer review of this work. Primary Handling Editor: Madhura Mukhopadhyay, in collaboration with the Nature Methods team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Pericytes colocalize to capillaries.
a-c, Pericytes immunostained with NG2 (a, green), α-SMA (b, green), and PDGFR-β (c, green) and ECs immunostained with CD31 (magenta). (n = 3), scale bars, 30 μm.
Extended Data Fig. 2 ECs, pericytes, and fibroblast are essential for formation of dense and stable capillary-bed.
a, b, EC only bioink; c, d, EC + pericyte bioink; e, f, EC + fibroblasts bioink; g, h, EC + pericyte + fibroblasts bioink 7 days (a, c, e, g) and 10 days (b, d, f, h) after bioprinting. Tissues fixed at analyzed for GFP expression (green) (n = 4). Scale bars, 500 μm.
Extended Data Fig. 3 Time course of fenestration marker expression in 3D-oBRB.
a-d, 3D vascular growth within tissues fixed at week 1 (a), week 2 (b), week 3 (c), and week 4 (d). Tissues were immunostained with FELS (green) and CD31 (magenta). Scale bars, 50 μm. n = 3.
Extended Data Fig. 4 ECM protein expressions in 3D-oBRB.
a-f, 3D reconstructed images of tissues immunostained for CD31 (magenta, a-f), nuclei (blue, a-f), LAMININ (LMN, green a, b), ELASTIN (ELN, green, c, d), COLLAGEN IV (COL IV, green, e, f), viewed at the subRPE level (a, c, e) and choriocapillaris level (b, d, f). N = 3. g, cross section of 3D-oBRB immunostained for Bruch’s membrane proteins LAMININ (yellow) and COLLAGEN IV (COL IV, magenta). Nuclei stained with DAPI (cyan). N = 3. Scale bar, 10μm.
Extended Data Fig. 5 Time course study of dry AMD phenotype induction in 3D-oBRB.
a-l, Nile red (yellow), anti-CD31 (magenta), and Hoechst (nucleus, blue) stained en face images of RPE (a-f), RPE-proximal choriocapillaris (g-l), and RPE-distal choriocapillaris (m-r) from 3D-oBRB treated with complement incompetent human serum (CI-HS, a, c, e, g, i, k, m, o, q) complement competent human serum (CC-HS, b, d, f, h, j, l, n, p, r) for 2, 4, and 7 days. Scale bars, 60 μm (a-l), 300 μm (m-r) n = 3. s-x, Nile red (yellow), anti-CD31 (magenta), and Hoechst (nucleus, blue) stained lateral view 3D rendered images of 3D-oBRB, treated with CI-HS (s, u, w) or CC-HS (t, v, x).
Extended Data Fig. 6 ML228 treatment on 2D-iRPE and 3D-oBRB.
a-f, RPE monoculture at 48 hr (a,d), 96 hr (b,e), and 2 weeks (c,f) from the beginning of ML228 (2 µM; 96 hr) treatment, immunostained with HIF-1α (magenta), ZO-1 (green), and Hoeschst (blue). DMSO uused to dissolve ML228 was used as the vehicle. Scale bars, 30 µm. (n = 3) g, TER measurement of 2D-iRPE without or with ML228 treatment (n = 3). Data are presented as mean values +/− SEM. Two-way ANOVA, Tukey’s multiple comparisons test were used for statistical analysis. h, ZO-1 staining based morphometry analysis of individual cell area in vehicle and ML228-treated samples was performed, (n = 1644, number of cells). Data are presented as mean values with standard deviation. ANOVA and Tukey’s multiple comparisons test were used for statistical analysis.
Extended Data Fig. 7 Activation of HIF-1α in RPE induces type-I CNV-like phenotype in 3D-oBRB.
a-h, immunostaining of tissues with anti-CD31 (magenta), anti-HIF-1α (yellow) antibodies, and staining for F-ACTIN (cyan), and nuclei (blue). Tissues were treated with either vehicle (DMSO, a,e), or ML228 (2 μM) on apical side of RPE only (b,f), or basal side of choroid only (c,g), or both sides (d,h). Red arrowheads indicate type-I CNV-like phenotype, white arrowheads indicate examples of HIF-1α translocation to the nuclei. (n = 3), scale bars, 50 μm. i, TER measurements normalized to ctrl. (n = 6). Data are presented as mean values +/− SEM. ANOVA and Tukey’s multiple comparisons test. j, k, pseudo 3D-projected side views of 3D-oBRB immunostained with anti-CD31 (magenta), anti-COL IV (yellow), anti-LAMININ (cyan) antibodies, and nuclei (blue). Tissues were treated with either vehicle (DMSO, j), or ML228 (2 μM) on apical side of RPE only (k). Arrowheads indicate type-I CNV-like phenotype in ML228-treated (k) tissues with choriocapillaris (CC) penetrating through the Bruch’s membrane (BM). (n = 3), scale bars, 10 μm.
Extended Data Fig. 8 STAT3 overexpression in the RPE induces type-I CNV-like phenotype in 3D-oBRB.
a-l, en face views (a-h), and pseudo 3D-projected side views (i, j) of 3D-oBRB immunostained at week 6 with anti-CD31(magenta), anti-STAT3 (yellow) antibodies, and stained for nuclei (blue), containing wildtype iRPE (a-d, i), STAT3 overexpressing iRPE (e-h, j). BM – Bruch’s membrane, CC – choriocapillaris. N = 3. Scale bars, 50 μm (a-c, e-g), 10 μm (i, j). k, TER measurements normalized to control from 2D-iRPE monoculture. (N = 8). Data are presented as mean values +/− SEM. Unpaired t-test was used for statistical analysis.
Extended Data Fig. 9 Bevacizumab suppresses type-I CNV-like phenotype in 3D-oBRB.
a-c, Images of deep choroidal regions of (a) vehicle, (b) ML228, (c) ML228 + Bevacizumab treated 3D-oBRB, immunostained for CD31 (red) and stained for Hoechst (blue). Scale bars, 350 μm. (n = 4). d-h, en face views (d-g), and pseudo 3D-projected side views (h) of STAT3 overexpressing 3D-oBRB treated with Bevacizumab immunostained at week 6 with anti-CD31(magenta), anti-STAT3 (yellow) antibodies, and stained for nuclei (blue). BM – Bruch’s membrane, CC – choriocapillaris. N = 3. Scale bars, 50 μm (d-g), 10 μm (h). i, TER measurements normalized to control of STAT3 overexpressing 3D-oBRB and STAT3 overexpressing 3D-oBRB treated with Bevacizumab. (n = 4), Data are presented as mean values + /- SEM. One-way ANOVA and Tukey’s multiple comparisons test were used for statistical analysis.
Supplementary information
Supplementary information
Supplementary Tables 1–4, Supplementary Figs 1–21, legends for Supplementary Videos 1–7, Angiogenesis quantification MATLAB code, and Supplementary Protocol.
Supplementary Video 1
Bioprinting of vascularized tissue with GFP-positive ECs and VEGF-dependent angiogenesis from day 4 to day 6.
Supplementary Video 2
Pericytes and EC tubes in the context of 3D vascularized tissue at day 7.
Supplementary Video 3
3D-oBRB tissue model.
Supplementary Video 4
ELASTIN and LAMININ formation in 3D-oBRB tissue model (z-stack).
Supplementary Video 5
ELASTIN and LAMININ formation in 3D-oBRB tissue model.
Supplementary Video 6
Complement induced dry AMD model.
Supplementary Video/ Movie 7
HIF-1α induced CNV in 3D-oBRB with anti-VEGF (Bevacizumab) treatment.
Supplementary Data 1
Statistical source data for Supplementary Fig. 4.
Supplementary Data 2
Unprocessed gels or blots, unprocessed figure for Supplementary Fig. 21.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data
Source Data Fig. 6
Statistical source data and source data (Fig. 6.zip) is on Figshare (https://doi.org/10.6084/m9.figshare.21300198)
Source Data Extended Data Fig 6
Statistical source data.
Source Data Extended Data Fig 7
Statistical source data.
Source Data Extended Data Fig 8
Statistical source data.
Source Data Extended Data Fig 9
Statistical source data.
Rights and permissions
About this article
Cite this article
Song, M.J., Quinn, R., Nguyen, E. et al. Bioprinted 3D outer retina barrier uncovers RPE-dependent choroidal phenotype in advanced macular degeneration. Nat Methods 20, 149–161 (2023). https://doi.org/10.1038/s41592-022-01701-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-022-01701-1
This article is cited by
-
New Perspectives in Stem Cell Transplantation and Associated Therapies to Treat Retinal Diseases: From Gene Editing to 3D Bioprinting
Stem Cell Reviews and Reports (2024)
-
Setting standards for stem cells
Nature Methods (2023)