Abstract
Planar-chiral cyclophanes have gained considerable concerns for drug discovery due to their unique conformational strain and 3D structure. However, the enantioselective synthesis of planar-chiral cyclophanes is a long-standing challenge for the synthetic community. We herein describe an N-heterocyclic carbene (NHC)-catalyzed asymmetric construction of planar-chiral cyclophanes. This transformation occurs through a dynamic kinetic resolution (DKR) process to convert racemic substrates into planar-chiral macrocycle scaffolds in good to high yields with high to excellent enantioselectivities. The ansa chain length and aromatic ring substituent size is crucial to achieve the DKR approach. Controlled experiments and DFT calculations were performed to clarify the DKR process.
Similar content being viewed by others
Introduction
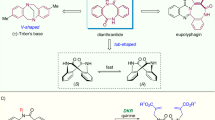
Macrocycles exhibit unique properties including shape diversity, conformational pre-organization, and conformational flexibility, compared to small-sized rings, particularly, five- to seven-membered rings1,2. The large surface area and tunable conformation of macrocycles increase the likelihood that the macrocycle will make meaningful contact with a biological target1,3. The compilation of macrocyclic small molecule screening libraries is critical to success when challenging, underexploited, or poorly “druggable” biological targets, such as lorlatinib, are involved1,4. Cyclophanes, which are molecules with an aromatic scaffold bearing a cross-linked chain, also called an ansa chain, are a subset of macrocycles5,6,7. Notably, cyclophane with a short ansa chain and bulky aromatic substituents exhibit planar chirality, which arises from the restricted bond flip of the aromatic ring8,9,10. Although planar-chiral cyclophanes are part of small molecule screening collections, these compounds have been increasingly used in drug development11,12, asymmetric synthesis13, and functional materials14 (Fig. 1A), and additional planar-chiral cyclophanes could benefit these collections. However, the enantioselective synthesis of planar-chiral cyclophanes represents a formidable challenge to the synthetic community and hampers their widespread inclusion in drug development efforts15,16.
Historically, the asymmetric synthesis of cyclophanes relied on chiral pools and chiral auxiliaries17,18,19,20,21. Asymmetric catalysis now provides access to the enantiopure planar-chiral cyclophanes16, as seen in Tanaka’s seminal asymmetric arene formation strategy to produce chiral metacyclophanes via rhodium-catalyzed alkyne cyclotrimerization22. Inspired by Tanaka’s pioneering work, several elegant enantioselective inter- and intramolecular [2 + 2 + 2] cycloaddition processes were reported for planar-chiral cyclophane assembly (Fig. 1B, I)23,24,25. In addition, asymmetric macrocyclization strategies have also provided access to planar-chiral cyclophanes26,27,28,29,30,31,32. Very recently, Collins28, Yang29, and Li30 independently developed enzyme-, chiral phosphoric acid (CPA)- or Pd-catalyzed asymmetric macrocyclization to achieve planar-chiral macrocycles (Fig. 1B, II). In addition to these processes, the desymmetrization is also an attractive method to afford planar-chiral structures. Shibata achieved the enantioselective synthesis of planar-chiral cyclophanes by ortho-lithiation of the achiral cyclophanes (Fig. 1B, III)33. The fourth is dynamic kinetic resolution (DKR) of achiral cyclophanes (Fig. 1B, IV). The challenges in generating planar-chiral cyclophanes by a DKR process involve identifying appropriately sized aromatic ring substituents and ansa chain lengths, which will allow rapid racemization of the substrate but limit the bond flip of the aromatic ring in the product. Both components are necessary to ensure the configurational stability of the resulting planar-chiral cyclophanes. In this scenario, Shibata was able to produce the chiral cyclophanes by a double asymmetric Sonogashira coupling of achiral [n, n]paracyclophanes34. More recently, Yang demonstrated an elegant CPA-catalyzed asymmetric electrophilic aromatic amination protocol to deliver planar-chiral macrocycles35.
Despite the previously mentioned straightforward approaches to afford planar-chiral cyclophanes, limitations include variable enantiomeric excess for different substrates, low reaction efficiencies, limited scope, and the use of stoichiometric amounts of chiral reagents. Further, the current protocols are restricted to a handful of catalytic models, and organocatalytic enantioselective synthetic routes are still in their infancy16. Consequently, these challenges prompted us to develop a DKR approach to achieve optically pure planar-chiral cyclophanes.
Chiral N-heterocyclic carbenes (NHCs) have been widely used as powerful organo-catalysts to access molecular architectures36,37,38,39,40,41,42,43,44. However, most previous reports focused on the synthesis of central chirality, and more recently, axial chirality45,46,47,48,49. Only recently has successful control of planar-chiral ferrocenes through NHC catalysis been reported by Chi50. Herein, we disclose an NHC-catalyzed DKR of racemic cyclophanes by an oxidative esterification reaction to afford the planar-chiral cyclophanes in good to high yields with high to excellent enantioselectivities (Fig. 1C). We have also demonstrated that our enantioenriched products can be easily converted into diverse planar-chiral scaffolds for potential library inclusion and asymmetric catalysis through cross-coupling reactions. Controlled experiments and DFT calculations were performed to clarify the DKR process.
Results
Reaction optimization
We set out to explore the DKR of cyclophanes by examining the reaction of [14]paracyclophane 1a with (3,5-di-tert-butylphenyl)methanol 2a in the presence of triazolium NHC as catalyst (Fig. 2). The atropisomerism of 1a was observed by variable temperature 1H NMR experiments (below −50 °C), suggesting a low rotational energy barrier for [14]cyclophane 1a at room temperature. Using 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) as base, 3,3’,5,5’-tetratert-butyldiphenoquinone (DQ) as oxidant, and THF as solvent, various chiral triazoline NHC catalysts I–VII were initially screened (entries 1–7). The pyrrolotriazolium-derived NHC catalyst I exhibited high conversion but low enantioselectivity, while amino alcohol-derived chiral NHC VII afforded the desired product in good yield with moderate enantioselectivity (entries 1 and 7). The N-substituents of the indanol-derived catalysts have an obvious impact on the enantioselectivity of the reaction, as N-2,4,6-(Me)3C6H2 (Mes), N-2,4,6-(Br)3C6H2, or N-C6F5 substituents gave the product in high yield but with poor enantioselectivity (entries 2, 3, and 6). Pleasingly, the enantioselectivity was dramatically increased when bulky N-2,4,6-(iPr)3C6H2 or N-2,4,6-Cy3C6H251 substituted indanol-derived NHC IV or V catalyst was used (entry 4–5, 93:7 er). Further base optimization indicated that strong bases, such as DBU and Cs2CO3, resulted in higher yields compared to relatively weak bases (entries 8–10). Subsequent solvent experimentation demonstrated that THF achieved the highest yields (entries 11–13). Decreasing the catalyst loading was not detrimental to reaction outcomes (entry 14). Interestingly, increase of temperature yielded the product in a higher er (entry 15). The improved enantioselectivity might arise from a faster racemization rate of the substrate, which facilitate the DKR process. The absolute configuration of 3aa was unambiguously determined by X-ray diffraction analysis of its derivative (vide infra).
aReaction conditions: A mixture of 1a (0.10 mmol), 2a (0.12 mmol), DQ (0.10 mmol), Base (10 mol%), and NHC catalyst (10 mol%) in solvent (2.0 mL) was stirred at room temperature for 24 h, the number in the black circle is the ansa chain length. bIsolated yield. cDetermined by HPLC using a chiral stationary phase. dNHC catalyst (5 mol%); the reaction was stirred for 48 h. eThe reaction was performed at 40 °C.
Substrate scope
With the optimized conditions in hand, we then evaluated the effect of the alcohol, starting with substituents on the phenyl ring of the benzyl alcohol. Regardless of the steric and electronics of the aromatic ring substituents, the desired planar-chiral cyclophanes (3ab–3ad) were afforded good yield with moderate to good enantioselectivities (Fig. 3). When substrates bearing bulkier groups were used, good yields and high enantioselectivities were achieved (3ae, 78% yield and 95:5 er). When other cyclic secondary alcohols were evaluated, the corresponding products 3af–3aj were obtained in 48–78% yield with er ranging from 93:7 – >99:1. Moreover, the benzyl alcohol with a 4-prenyl group was not an appropriate substrate for this reaction under the optimized conditions (3ak).
The investigation of ansa chain length and substituent size was conducted using 2e as model substrate (Fig. 4). Reducing to 11- or 12-membered ansa chains, the reaction went through a kinetic resolution process, in which planar-chiral products 3be and 3ce were obtained in 86:14 er and 91.5:8.5 er, respectively, while 1c was recovered with 51% yield and 82:18 er. Using a 13-membered ansa chain was feasible, delivering the planar-chiral [13]paracyclophane 3de with moderate yield and good enantioselectivity. Increasing the ansa chain length and using a 15-membered compound was tolerated and gave the product 3ee in 72% yield with a 95:5 er value, while [16]paracyclophane 3fe led to the loss of planar chirality due to the relative low rotation barrier.
aThe reaction was performed at room temperature, IV was used as catalyst. bThe reaction was performed at 40 °C, V was used as catalyst. c0.2 mmol of 2a was used. dThe reaction was performed at 50 °C, V was used as catalyst. eThe reaction was stirred at room temperature for 24 h, 1k was recovered in 55:45 er, and 1 l was recovered in 50:50 er.
In addition, an examination of the aromatic ring substituent size was performed (Fig. 4). 4-H substituted [14]paracyclophane resulted in the loss of planar chirality (3ge), but [14]paracyclophanes bearing chloro, methyl, or iodo groups on the benzene ring allowed the NHC-catalyzed DKR to occur, generating the planar-chiral cyclophanes in good yields and enantioselectivities (3he–3je). Installment of phenyl or ethynyl groups on the aromatic ring was also tolerated. The products 3ke and 3le were obtained in high er value, and corresponding substrates 2k and 2l were recovered in low er value, which suggested racemization of the substrates occurred. These results indicate that the DKR of planar-chiral [n]paracyclophanes is highly dependent on ansa chain length and the size of substituents in the aromatic ring, which allows substrate racemization but limits bond flip in the product’s aromatic ring.
Encouraged by these results, we continued to explore the scope of [n]paracyclophanes in this NHC-catalyzed DKR reaction. A diverse array of achiral paracyclophanes with a variety of ansa chain lengths and substituents were tested (Fig. 5). [13]paracyclophane with a chloro group at the 4-position of the phenyl ring performed well to deliver the corresponding planar chiral product (5ae) in good yield and enantioselectivity. [14] and [15]paracyclophanes bearing different substitutes (vinyl, iodo, ethynyl, phenyl) at the phenyl ring were also accommodated and provided the enantioenriched products (5be–5fe). Heteroaromatic rings like 3-thienyl could be introduced at the 4-position of the substrates (5ge, 5he). [16] and [17]paracyclophane with 2-naphthyl, benzofuran-6-yl or benzo[b]thiophen-3-yl yielded the produts in high enantioselectivity (5ie–5ke), as could [18]paracyclophane with benzofuran-6-yl albeit with lower enantioselectivity (5le). In addition, modifications of the ansa chain were also investigated. The yield of [14] and [15]paracyclophane with an ester group or nitrogen-linked ansa chain decreased to 26% and 16% (5me, 5ne), respectively. The decreased yield of 5me probably originated from substrate decomposition under the reaction conditions. [15]paracyclophane with an oxygen-linked ansa chain afforded the product in good yield with high enantioselectivity (5oe). To assess the configurational stability of the planar-chiral products, 3ae in toluene was heated to 110 °C. Notably, HPLC analysis indicated that no racemization of 3ae was observed, even after 7 days at this temperature (see Supplementary Information).
To expand the potential utility of this process, we further manipulated the planar-chiral cyclophane (Fig. 6A). The ester in 3ae could be reduced with LiAlH4 to generate benzylic alcohol 6 in 98% yield with only a slight erosion of enantioselectivity. In addition, a Ni-catalyzed cross–coupling reaction of 3ae with diphenylphosphine afforded planar-chiral phosphine 7 in 61% yield with a slight loss of stereochemical integrity due to the vigorous reaction conditions. Moreover, the enantiomerically enriched triazole 8 was obtained by a Sonogashira cross–coupling, desilication, and CuTC-catalyzed click reaction sequence. Finally, 3ae was easily transferred to corresponding pyrene-bearing product 9 in 68% yield and 93:7 er through a Suzuki–Miyara cross–coupling reaction with pyren-1-ylboronic acid. The absolute configuration of 10 was unambiguously determined by X-ray diffraction analysis (https://www.ccdc.cam.ac.uk/data_request/cif) (Fig. 6B).
A Synthetic transformation of enantioenriched [14]paracyclophane 3ae. Reaction conditions: (i) LiAlH4, 0 °C, THF, 2 h. (ii) Ni(dppe)Cl2, Ph2PH, Et3N, DMF, 120 °C, 12 h. (iii) Pd(PPh3)2Cl2, CuI, TMS-acetylene, Et3N, 50 °C, 10 h, THF, 91% yield, 93:7 er. (iv) TBAF•3H2O, THF, rt, 4 h, 87% yield, 93:7 er. (v) CuTC, (R)-Camphor-10-sulfonyl azide, toluene, rt, 4 h, 96% yield, 13:1 dr. (vi) Pd(PPh3)4, Na2CO3, pyren-1-ylboronic acid, 1,4-dioxane, 100 °C, 24 h, 68% yield, 93:7 er. B X-ray of planar-chiral cyclophane 10.
A postulated mechanism is illustrated in Fig. 7. The reaction starts from the addition of NHC catalyst IV to aldehyde 1a to generate Breslow intermediate Int-I. Then, the oxidation of Int-I forms acylazolium intermediate Int-II. Finally, the esterification of Int-II with benzylic alcohol 2e delivers the planar-chiral product 3ae and releases the free NHC catalyst IV.
The DKR process originates from the rapid bond flip of the aromatic ring in the two enantiomers ([Sp]−1a and [Rp]−1a) and the reaction-rate difference with the addition of NHC catalyst (Fig. 8, eq 1). To clarify the DKR process, several controlled experiments were conducted. The reaction of [15]paracyclophane 4f with 2e was performed under standard conditions for 12 h; 5fe was obtained in 48% yield with 90:10 er, and 4f was recovered as racemates in 40% yield. The result indicates that the racemization of 4k is rapid and not the rate-determined step (Fig. 8, eq 2). In addition, as illustrated in the investigation of substrate scope, [12]paracyclophane underwent kinetic resolution, thus, enantioenriched 4-phenyl-substituted [12]paracyclophane (Rp)−11 was treated under the optimized reaction conditions, the reaction proceeds smoothly to deliver (Rp)−12 in 82% yield with >99:1 er value (Fig. 8, eq 3). Alternatively, only a trace amount of (Rp)−12 was obtained by using ent-IV as NHC catalyst (<5% yield), and the (Rp)−11 was recovered in 89% yield with 97:3 er value (Fig. 8, eq 4). Further, the reaction of 1a and 2e with ent-IV as catalyst gave a reverse selectivity, suggesting that the (Rp)−11 was unmatched with ent-IV (Fig. 8, eq 5). These results indicate that the reaction of NHC catalyst IV to (Rp)-11 is kinetically favored.
To further explore the energy-difference for the addition of NHC catalyst IV to the enantiomers (Sp)-1a and (Rp)-1a, a density functional theory (DFT) study was performed using Gaussian 09. As illustrated in Fig. 9, the energy barrier of the addition of NHC catalyst IV to (Rp)-1a is 3.6 kcal/mol lower than that of (Sp)-1a; thus, the addition of NHC catalyst IV to (Rp)-1a is kinetically favored. Further analysis of transition states TS-(Sp)-1a and TS-(Rp)-1a using atoms-in-molecules52,53 and noncovalent interactions analyses54 reveal that the weak interactions in TS-(Rp)-1a are stronger than those observed in TS-(Sp)-1a, which leads to TS-(Rp)-1a having a lower energy barrier. Interactions in TS-(Rp)-1a include: seven C–H…π interactions, four C–H…O hydrogen bond interactions, two C–H…N hydrogen bond interactions, one Lp…π interaction, one C–H…Br halogen bond interaction, and ten C–H…H–C van der Waals interactions. Interactions in TS-(Sp)-1a include: four C–H…π interactions, three C–H…O hydrogen bond interactions, one C–H…N hydrogen bond interaction, two C–H…Br halogen bond interactions, and nine C-H…H–C van der Waals (See Supplementary Fig. 2). Although the calculated results are consistent with the observed experiments, a relatively low Gibbs free energy for addition of NHC catalyst to the substrate suggests a rapid rate for this transformation. While the subsequent oxidative esterification is not the enantioselectivity-determined step, together with experimental obversion that increases the reaction temperature led to a higher enantioselectivity. Thus, the Curtin-Hammet scenario might be operative for the DKR approach55.
Discussion
In summary, we developed an NHC-catalyzed enantioselective synthesis of planar-chiral cyclophanes. The reaction features a DKR process involving an aldehyde oxidation esterification and affords a wide range of planar-chiral macrocycles in good to high yields with high enantioselectivities. An investigation of ansa chain length and the size of aromatic ring substituents indicated that these variables were crucial to generating the planar chirality. Further, the cyclophane products could be transformed into other planar-chiral macrocyclic scaffolds by simple reactions. Controlled experiments and DFT calculations were performed to clarify the DKR process. An application of these planar-chiral scaffolds for library inclusion and asymmetric catalysis are under investigation in our lab.
Methods
Materials. For 1H NMR, 13C NMR, and high-performance liquid chromatography spectra of compounds in this manuscript, see Supplementary Figures. For details of the synthetic macrocycle substrates: diphenol (10.0 mmol) and dibromides (10.0 mmol) in DMF (25.0 mL) were slowly added to a suspension of K2CO3 (3.45 g, 25.0 mmol) and NaI (170 mg, 1.0 mmol) in DMF (150 mL) at 140 °C over 18 h. The solvent was removed under reduced pressure. The aqueous phase was extracted with EtOAc (3 × 50 mL), and the combined organic layers were washed with water and brine, and dried over Na2SO4. The solvent was removed under reduced pressure, and the residue was purified by flash column chromatography on silica gel affording the products (yield: 10 ~ 40%).
Synthesis of 3 and 5. To a 15 mL Schlenk tube equipped with a magnetic stirring bar was added macrocycles substrates 1 or 4 (0.10 mmol), 2 (0.4 mmol), NHC precursor IV or V (0.005 mmol), DBU (0.01 mmol) and DQ (0.1 mmol). The tube was closed with a septum, evacuated, and refilled with nitrogen (3 cycles). Then, freshly distilled THF (1.0 mL) was added to the reaction mixture and stirred for 10–72 h. Upon completion (monitored by TLC), the solvent was evaporated, and the residue was purified by silica gel column chromatography to afford the planar-chiral cyclophanes 3 or 5.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The authors declare that the data supporting the findings of this study are available within the article and Supplementary Information file, or from the corresponding author upon request. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers CCDC 2256859 (for (Rp)-10). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. The full experimental details for the preparation of all new compounds, and their spectroscopic and chromatographic data generated in this study are provided in the Supplementary Information/Source Data file. All data are available from the authors upon request. Source data are provided with this paper.
References
Ciardiello, J. J., Stewart, H. L., Sore, H. F., Galloway, W. R. J. D. & Spring, D. R. A novel complexity-to-diversity strategy for the diversity-oriented synthesis of structurally diverse and complex macrocycles from quinine. Bioorg. Med. Chem. 25, 2825–2843 (2017).
Driggers, E. M., Hale, S. P., Lee, J. & Terrett, N. K. The exploration of macrocycles for drug discovery-an underexploited structural class. Nat. Rev. Drug Discov. 7, 608–624 (2008).
Lau, Y. H. et al. Double strain-promoted macrocyclization for the rapid selection of cell-active stapled peptides. Angew. Chem. Int. Ed. 54, 15410–15413 (2015).
Syed, Y. Y. Lorlatinib: first global approval. Drugs 79, 93–98 (2019).
Vögtle, F. & Neumann, P. Zur nomenklatur der phane. Tetrahedron Lett. 10, 5329–5334 (1969).
Cram, D. J. & Cram, J. M. Cyclophane chemistry: bent and battered benzene rings. Acc. Chem. Res. 4, 204–213 (1971).
Tobe, Y. & Sonoda, M. In Modern Cyclophane Chemistry, 1–40 (Wiley-VCH, Weinheim, 2004).
Marshall, J. A. Trans-cycloalkenes and [a.b]betweenanenes, molecular jump ropes and double bond sandwiches. Chem. Soc. Rev. 11, 213–218 (1980).
Liu, Z. C., Nalluri, S. K. M. & Stoddart, J. F. Surveying macrocyclic chemistry: from flexible crown ethers to rigid cyclophanes. Chem. Soc. Rev. 46, 2459–2478 (2017).
Tanaka, K. Catalytic enantioselective synthesis of planar chiral cyclophanes. Bull. Chem. Soc. Jpn. 91, 187–194 (2018).
Gulder, T. & Baran, P. S. Strained cyclophane natural products: macrocyclization at its limits. Nat. Prod. Rep. 29, 899–934 (2012).
Romero, D. Benefit with daratumumab maintenance. Nat. Rev. Clin. Oncol. 18, 676 (2021).
Zahid, H., Eduard, S., Knoll, D. M., Joerg, L. & Stefan, B. Planar chiral [2.2] paracyclophanes: from synthetic curiosity to applications in asymmetric synthesis and materials. Chem. Soc. Rev. 47, 6947–6963 (2018).
Ramaiah, D., Neelakandan, P. P., Nair, A. K. & Avirah, R. R. Functional cyclophanes: promising hosts for optical biomolecular recognition. Chem. Soc. Rev. 42, 4158–4168 (2010).
Sambasivarao, K., Eknath, S. M. & Waghule, G. T. Selected synthetic strategies to cyclophanes. Beilstein J. Org. Chem. 11, 1274–1331 (2015).
López, R. & Palomo, C. Planar chirality: a mine for catalysis and structure discovery. Angew. Chem. Int. Ed. 61, e202113504b (2022).
Kanomata, N. & Nakata, T. A compact chemical miniature of a holoenzyme, coenzyme nadh linked dehydrogenase. Design and synthesis of bridged nadh models and their highly enantioselective reduction1. J. Am. Chem. Soc. 122, 4563–4568 (2000).
Kanomata, N. & Ochiai, Y. Stereocontrol of molecular jump-rope: crystallization-induced asymmetric transformation of planar-chiral cyclophanes. Tetrahedron Lett. 42, 1045–1048 (2001).
Kanomata, N. & Oikawa, J. Adsorption-induced asymmetric transformation of planar-chiral pyridinophanes. Tetrahedron Lett. 44, 3625–3628 (2003).
Yoshida, Y., Kanashima, Y., Mino, T. & Sakamoto, M. Asymmetric syntheses and applications of planar chiral hypervalent iodine(v) reagents with crown ether backbones. Tetrahedron 75, 3840–3849 (2019).
Ueda, T., Kanomata, N. & Machida, H. Synthesis of planar-chiral paracyclophanes via samarium(II)-catalyzed intramolecular pinacol coupling. Org. Lett. 7, 2365–2368 (2005).
Tanaka, K., Sagae, H., Toyoda, K., Noguchi, K. & Hirano, M. Enantioselect syn planar chiral metacyclophane through Rh-cat alkyne cyclotrimerization [a,w-diyne rxn DMAD to bicyclic phthalate]. J. Am. Chem. Soc. 129, 1522–1523 (2007).
Tanaka, K., Sagae, H., Toyoda, K. & Hirano, M. Enantioselective synthesis of planar-chiral metacyclophanes through cationic Rh(I)/modified-BINAP-catalyzed inter- and intramolecular alkyne cyclotrimerizations. Tetrahedron 64, 831–846 (2008).
Araki, T., Noguchi, K. & Tanaka, K. Enantioselective synthesis of planar-chiral garba-paracyclophanes: rhodium-catalyzed [2 + 2 + 2] cycloaddition of cyclic diynes with terminal monoynes. Angew. Chem. Int. Ed. 52, 5617–5621 (2013).
Nogami, J. et al. Enantioselective synthesis of planar chiral zigzag-type cyclophenylene belts by rhodium-catalyzed alkyne cyclotrimerization. J. Am. Chem. Soc. 142, 9834–9842 (2020).
Tanaka, K., Hori, T., Osaka, T., Noguchi, K. & Hirano, M. Rhodium-catalyzed reactions of dithiols and 1,4-bis(bromomethyl)benzenes leading to enantioenriched dithiaparacyclophanes. Org. Lett. 9, 4881–4884 (2007).
Ding, Q., Wang, Q., He, H. & Cai, Q. Asymmetric synthesis of (−)-pterocarine and (−)-galeon via chiral phase transfer-catalyzed atropselective formation of diarylether cyclophane skeleton. Org. Lett. 19, 1804–1807 (2017).
Gagnon, C. et al. Biocatalytic synthesis of planar chiral macrocycles. Science 367, 917–921 (2020).
Yu, S., Shen, G., He, F. & Yang, X. Asymmetric synthesis of planar-chiral macrocycles via organocatalyzed enantioselective macrocyclization. Chem. Commun. 58, 7293–7296 (2022).
Wei, S., Chen, L.-Y. & Li, J. Enantioselective synthesis of planar chiral macrocyclic metacyclophanes by Pd-catalyzed C–O cross-coupling. ACS Catal. 13, 7450–7456 (2023).
Tan, L., Sun, M., Wang, H., Kim, J. & Lee, M. Enantiocontrolled macrocyclization by encapsulation of substrates in chiral capsules. Nat. Synth. 2, 1222–1231 (2023).
Yang, G., He, Y., Wang, T. Li, Z. & Wang, J. Atroposelective synthesis of planar-chiral indoles via carbene catalyzed macrocyclization. Angew. Chem. Int. Ed. 63, e202316739 (2024).
Kanda, K., Endo, K. & Shibata, T. Enantioselective synthesis of planar-chiral 1, n-dioxa[n]paracyclophanes via catalytic asymmetric ortho-lithiation. Org. Lett. 12, 1980–1983 (2010).
Kanda, K., Koike, T., Endo, K. & Shibata, T. The first asymmetric sonogashira coupling for the enantioselective generation of planar chirality in paracyclophanes. Chem. Commun. 14, 1870–1872 (2009).
Wang, D., Shao, Y.-B., Chen, Y., Xue, X.-S. & Yang, X. Y. Enantioselective synthesis of planar-chiral macrocycles through asymmetric electrophilic aromatic amination. Angew. Chem. Int. Ed. 61, e202201064 (2022).
Nair, V. et al. Employing homoenolates generated by NHC catalysis in carbon–carbon bond-forming reactions: state of the art. Chem. Soc. Rev. 40, 5336–5346 (2011).
Biju, A. T., Kuhl, N. & Glorius, F. Extending NHC-catalysis: coupling aldehydes with unconventional reaction partners. Acc. Chem. Res. 44, 1182–1195 (2011).
Bugaut, X. & Glorius, F. Organocatalytic umpolung: N-heterocyclic carbenes and beyond. Chem. Soc. Rev. 41, 3511–3522 (2012).
Ryan, S. J., Candish, L. & Lupton, D. W. Cheminform abstract: acyl anion free N‐heterocyclic carbene organocatalysis. Chem. Soc. Rev. 42, 4906–4917 (2013).
Mahatthananchai, J. & Bode, J. W. On the mechanism of N-heterocyclic carbene-catalyzed reactions involving acyl azoliums. Acc. Chem. Res. 47, 696–707 (2014).
Hopkinson, M. N., Richter, C., Schedler, M. & Glorius, F. An overview of N-heterocyclic carbenes. Nature 510, 485–496 (2014).
Ishii, T., Nagao, K. & Ohmiya, H. Recent advances in N-heterocyclic carbene-based radical catalysis. Chem. Sci. 11, 5630–5636 (2020).
Chen, X.-K., Wang, H.-L., Jin, Z.-C. & Chi, Y. R. N-heterocyclic carbene organocatalysis: activation modes and typical reactive intermediates. Chin. J. Chem. 38, 1167–1202 (2020).
Chen, X.-Y., Cao, Z.-H. & Ye, S. Bifunctional N-heterocyclic carbenes derived from i-pyroglutamic acid and their applications in enantioselective organocatalysis. Acc. Chem. Res. 53, 690–702 (2020).
Feng, J. & Du, D. Asymmetric synthesis of atropisomers enabled by N-heterocyclic carbene catalysis. Tetrahedron 100, 132456 (2021).
Song, R., Xie, Y., Jin, Y. & Chi, Y. R. Carbene-catalyzed asymmetric construction of atropisomers. Angew. Chem. Int. Ed. 60, 26026–26037 (2021).
Wang, J. M., Zhao, C. G. & Wang, J. Recent progress toward the construction of axially chiral molecules catalyzed by an N-heterocyclic carbene. ACS Catal. 11, 12520–12531 (2021).
Lu, S., Poh, S. B. & Zhao, Y. Kinetic resolution of 1,1’-biaryl-2,2′-diols and amino alcohols through NHC-catalyzed atroposelective acylation. Angew. Chem. Int. Ed. 46, 11041–11045 (2015).
Zhao, C. G. et al. Enantioselective [3 + 3] atroposelective annulation catalyzed by N-heterocyclic carbenes. Nat. Commun. 9, 611 (2018).
Lv, X. K. et al. Access to planar chiral ferrocenes via N-heterocyclic carbene-catalyzed enantioselective dsymmetrization reactions. ACS Catal. 12, 2706–2713 (2022).
Yan, J.-L. et al. Carbene-catalyzed atroposelective synthesis of axially chiral styrenes. Nat. Commun. 13, 84 (2022).
McLean, A. & Chandler, D. G. S. Contracted gaussian basis sets for molecular calculations. I. second row atoms, Z = 11–18. J. Chem. Phys. 72, 5639–5648 (1980).
Raghavachari, K., Binkley, J. S., Seeger, R. & Pople, J. A. Self‐consistent molecular orbital methods. XX. A basis set for correlated wave functions. J. Chem. Phys. 72, 650–654 (1980).
Bader, R. F. W. Atoms in molecules. Acc. Chem. Res. 18, 9–15 (1985).
Seeman, J. I. Effect of conformational change on reactivity in organic chemistry. evaluations, applications and extensions of Curtin−Hammett Winstein−Holness kinetics. Chem. Rev. 83, 83–134 (1983).
Acknowledgements
This project was financially supported by the National Natural Science Foundation of China (No. 22171027, 22103005), the Beijing Natural Science Foundation (No. 2212009), the Fundamental Research Funds for the Central Universities (No. 2233300007) and the High Performance Computing Platform of Beijing University of Chemical Technology (BUCT). We thank Prof. Dr. Guofu Zi (Beijing Normal University) for the X-ray diffraction analysis and Dr. Stephanie A. Blaszczyk for proofread the manuscript (Moxie Medical Writing).
Author information
Authors and Affiliations
Contributions
J.Y.L. conducted main experiments; Z.Y.D. prepared several starting materials; X.E.Y. optimized the reaction conditions and prepared several starting materials during the revision of the manuscript. Y.C.and Z.H.Y. performed part of the DFT computation; M.W. and C.Y.L. rerun the computation during the revision of the manuscript. C.G.Z. conceptualized and directed the project, and drafted the manuscript with the assistance from co-authors. All authors contributed to discussions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Reprints and permission information is available online at http://npg.nature.com/reprintsandpermissions/
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, J., Dong, Z., Chen, Y. et al. N-Heterocyclic carbene-catalyzed enantioselective synthesis of planar-chiral cyclophanes via dynamic kinetic resolution. Nat Commun 15, 2338 (2024). https://doi.org/10.1038/s41467-024-46376-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-46376-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.