Featured
-
-
Article
| Open Access TANGO6 regulates cell proliferation via COPI vesicle-mediated RPB2 nuclear entry
TANGO6 regulates cell proliferation via COPI vesicle-mediated RPB2 nuclear entry
How RNA polymerase II subunits enter the nucleus is not well understood. Here, the authors show that Transport and Golgi organization protein 6, TANGO6, recruits RNA polymerase II subunit B2, RPB2, to the ER membrane in a retrograde manner and transports it to the nucleus with the aid of importins.
- Zhi Feng
- , Shengnan Liu
- & Li Li
-
Article
| Open Access piRNA loading triggers MIWI translocation from the intermitochondrial cement to chromatoid body during mouse spermatogenesis
piRNA loading triggers MIWI translocation from the intermitochondrial cement to chromatoid body during mouse spermatogenesis
piRNA processing factors and piRNA/PIWI complex are found in intermitochondrial cement (IMC) and chromatoid body (CB). Here, the authors show that piRNA loading orchestrates MIWI translocation from the IMC to CB, demonstrating its crucial role in spermatogenesis.
- Huan Wei
- , Jie Gao
- & Mo-Fang Liu
-
Article
| Open Access Simultaneous proteome localization and turnover analysis reveals spatiotemporal features of protein homeostasis disruptions
Simultaneous proteome localization and turnover analysis reveals spatiotemporal features of protein homeostasis disruptions
Protein function depends on their subcellular location and turnover rate. Here, the authors report a method to measure spatial and temporal proteome dynamics simultaneously, revealing compartment-specific protein turnover and translocation in cardiac cells under ER stress and carfilzomib treatment.
- Jordan Currie
- , Vyshnavi Manda
- & Edward Lau
-
Article
| Open Access The GET insertase exhibits conformational plasticity and induces membrane thinning
The GET insertase exhibits conformational plasticity and induces membrane thinning
Tail-anchored (TA) membrane protein biogenesis is mediated by the GET insertase. Here, authors present cryo-EM and X-ray structures, MD simulations and functional data for human and fungal insertases showing membrane remodeling for TA insertion.
- Melanie A. McDowell
- , Michael Heimes
- & Irmgard Sinning
-
Article
| Open Access Excessive copper impairs intrahepatocyte trafficking and secretion of selenoprotein P
Excessive copper impairs intrahepatocyte trafficking and secretion of selenoprotein P
Selenium and copper are two essential trace elements whose homeostasis and distribution is regulated by hepatic release of selenoprotein P (SELENOP) and ceruloplasmin, respectively. Here, the authors show that excessive copper results in hepatic SELENOP accumulation in the trans Golgi which might limit the selenium transport to peripheral organs.
- Maria Schwarz
- , Caroline E. Meyer
- & Anna P. Kipp
-
Article
| Open Access Orthodenticle homeobox 2 is transported to lysosomes by nuclear budding vesicles
Orthodenticle homeobox 2 is transported to lysosomes by nuclear budding vesicles
Many homeodomain transcription factors are secreted and move to neighboring cells. Here, orthodenticle homeobox 2 is shown to be exported from the nucleus in a nuclear membrane, which buds off to then be degraded or secreted.
- Jun Woo Park
- , Eun Jung Lee
- & Jin Woo Kim
-
Article
| Open Access Barrier properties of Nup98 FG phases ruled by FG motif identity and inter-FG spacer length
Barrier properties of Nup98 FG phases ruled by FG motif identity and inter-FG spacer length
The permeability barrier of the nuclear pore assembles from cohesive FG repeats. By systematic engineering and testing repeat variants, the authors pinpointed the sequence features that rule barrier assembly and transport selectivity.
- Sheung Chun Ng
- , Abin Biswas
- & Dirk Görlich
-
Article
| Open Access Hedgehog is relayed through dynamic heparan sulfate interactions to shape its gradient
Hedgehog is relayed through dynamic heparan sulfate interactions to shape its gradient
The Hedgehog morphogen creates gradients during development, but diffusion alone cannot explain its spatiotemporal dynamics. Hedgehog transport requires binding heparan sulfate sugar chains, and the authors now show that Hedgehogs can spread by interacting with sequential heparan molecules.
- Fabian Gude
- , Jurij Froese
- & Kay Grobe
-
Article
| Open Access The mitochondrial Hsp70 controls the assembly of the F1FO-ATP synthase
The mitochondrial Hsp70 controls the assembly of the F1FO-ATP synthase
The mitochondrial ATP synthase produces the bulk of cellular ATP. Here, the authors report a function of the mitochondrial Hsp70 in the formation of the catalytical head and in its assembly with the peripheral stalk to form the mature ATP synthase.
- Jiyao Song
- , Liesa Steidle
- & Thomas Becker
-
Article
| Open Access Protein import motor complex reacts to mitochondrial misfolding by reducing protein import and activating mitophagy
Protein import motor complex reacts to mitochondrial misfolding by reducing protein import and activating mitophagy
Mitophagy activation is mediated by mitochondrial depolarization. Here, the authors show that mitochondrial protein misfolding can activate mitophagy in a depolarization-independent manner mediated by a protein import reduction.
- Jonas Benjamin Michaelis
- , Melinda Elaine Brunstein
- & Christian Münch
-
Article
| Open Access Stomatin modulates adipogenesis through the ERK pathway and regulates fatty acid uptake and lipid droplet growth
Stomatin modulates adipogenesis through the ERK pathway and regulates fatty acid uptake and lipid droplet growth
Stomatin is a component of lipid rafts. Here, Wu et al. show that stomatin modulates the differentiation and functions of adipocytes by regulating adipogenesis signaling and fatty acid influx such that with excessive calorie intake, increased stomatin induces adiposity.
- Shao-Chin Wu
- , Yuan-Ming Lo
- & Chi-Hung Lin
-
Article
| Open Access Ribosome profiling reveals multiple roles of SecA in cotranslational protein export
Ribosome profiling reveals multiple roles of SecA in cotranslational protein export
Using a combination of ribosome profiling methods, Zhu et al. investigate the principles governing the cotranslational interaction of SecA with nascent proteins and reveal a hierarchical organization of protein export pathways in bacteria.
- Zikun Zhu
- , Shuai Wang
- & Shu-ou Shan
-
Article
| Open Access Mechanical control of nuclear import by Importin-7 is regulated by its dominant cargo YAP
Mechanical control of nuclear import by Importin-7 is regulated by its dominant cargo YAP
The translation of mechanical cues into gene expression changes is dependent on the nuclear import of mechanoresponsive transcriptional regulators. Here the authors identify that Importin-7 drives the nuclear import of one such regulator YAP while YAP then controls Importin-7 response to mechanical cues and restricts Importin-7 binding to other cargoes.
- María García-García
- , Sara Sánchez-Perales
- & Miguel A. Del Pozo
-
Article
| Open Access Mapping protein interactions in the active TOM-TIM23 supercomplex
Mapping protein interactions in the active TOM-TIM23 supercomplex
The TOM and TIM23 complexes facilitate the transport of nuclear-encoded proteins into the mitochondrial matrix. Here, the authors use a stalled client protein to purify the translocation supercomplex and gain insight into the TOM-TIM23 interface and the mechanism of protein handover from the TOM to the TIM23 complex.
- Ridhima Gomkale
- , Andreas Linden
- & Peter Rehling
-
Article
| Open Access NEK1-mediated retromer trafficking promotes blood–brain barrier integrity by regulating glucose metabolism and RIPK1 activation
NEK1-mediated retromer trafficking promotes blood–brain barrier integrity by regulating glucose metabolism and RIPK1 activation
NEK1 mutations promote lethality early in life and ALS late in life via unknown mechanisms. Here, the authors show that NEK1 mutation disrupts retromer-mediated trafficking and promotes RIPK1 activation, connecting retromer trafficking and metabolism to neuroinflammation by dietary intervention.
- Huibing Wang
- , Weiwei Qi
- & Junying Yuan
-
Article
| Open Access Global kinome profiling reveals DYRK1A as critical activator of the human mitochondrial import machinery
Global kinome profiling reveals DYRK1A as critical activator of the human mitochondrial import machinery
Mitochondrial protein import is mediated by the translocase of the outer membrane (TOM), through which nearly all precursors traverse. Here, the authors perform global in vitro kinome profiling and by this identify that DYRK1A phosphorylates TOM70 and promotes import.
- Corvin Walter
- , Adinarayana Marada
- & Chris Meisinger
-
Article
| Open Access Recapitulation of selective nuclear import and export with a perfectly repeated 12mer GLFG peptide
Recapitulation of selective nuclear import and export with a perfectly repeated 12mer GLFG peptide
The permeability barrier of nuclear pore complexes blocks passage of inert macromolecules but allows rapid, receptor-mediated, and RanGTPase-driven transport of cargoes up to ribosome size. The authors now show that such a barrier can be faithfully recapitulated by an ultimately simplified FG phase assembled solely from a tandemly repeated 12mer GLFG peptide.
- Sheung Chun Ng
- , Thomas Güttler
- & Dirk Görlich
-
Article
| Open Access Control of membrane barrier during bacterial type-III protein secretion
Control of membrane barrier during bacterial type-III protein secretion
Type-III secretion systems (T3SSs) are capable of translocating proteins with high speed while maintaining the membrane barrier for small molecules. Here, a structure-function analysis of the T3SS pore complex elucidates the precise mechanisms enabling the gating and the conformational changes required for protein substrate secretion.
- Svenja Hüsing
- , Manuel Halte
- & Thibaud T. Renault
-
Article
| Open Access Thioesterase superfamily member 1 undergoes stimulus-coupled conformational reorganization to regulate metabolism in mice
Thioesterase superfamily member 1 undergoes stimulus-coupled conformational reorganization to regulate metabolism in mice
Cold exposure activates thermogenesis and fatty acid oxidation in brown fat, a process suppressed by Them1. Here, the authors show that cold induces Them1 phosphorylation and loss of puncta that suppress fatty acid use, leading to a diffuse localization and increased energy expenditure in mice.
- Yue Li
- , Norihiro Imai
- & Susan J. Hagen
-
Article
| Open Access Analysis of diverse eukaryotes suggests the existence of an ancestral mitochondrial apparatus derived from the bacterial type II secretion system
Analysis of diverse eukaryotes suggests the existence of an ancestral mitochondrial apparatus derived from the bacterial type II secretion system
Bacteria use the type 2 secretion system to secrete enzymes and toxins across the outer membrane to the environment. Here the authors analyse the T2SS pathway in three protist lineages and suggest that the early mitochondrion may have been capable of secreting proteins into the cytosol.
- Lenka Horváthová
- , Vojtěch Žárský
- & Pavel Doležal
-
Article
| Open Access LONP1 and mtHSP70 cooperate to promote mitochondrial protein folding
LONP1 and mtHSP70 cooperate to promote mitochondrial protein folding
Most mitochondrial proteins are imported from the cytosol and must fold in the mitochondria. Here, the authors show that the mitochondrial protease LONP1 plays a critical role in the mtHSP70 chaperone system independently of its protease activity.
- Chun-Shik Shin
- , Shuxia Meng
- & David C. Chan
-
Article
| Open Access A ribosome-associated chaperone enables substrate triage in a cotranslational protein targeting complex
A ribosome-associated chaperone enables substrate triage in a cotranslational protein targeting complex
Biochemistry combined with biophysical measurements and mathematical modeling offer insight into the mechanism by which the cotranslational chaperone, nascent polypeptide-associated complex (NAC), modulates substrate selection by signal recognition particle (SRP) and reduces aberrant, nonspecific targeting of ribosomes to the ER.
- Hao-Hsuan Hsieh
- , Jae Ho Lee
- & Shu-ou Shan
-
Article
| Open Access Concerted localization-resets precede YAP-dependent transcription
Concerted localization-resets precede YAP-dependent transcription
The transcriptional regulator YAP shuttles rapidly between the cytoplasm and nucleus, but whether and how dynamics such as amplitude and frequency affect target gene transcription is unclear. Here, using live imaging of endogenous YAP and target-gene transcription, the authors show that YAP-dependent signalling is encoded through rapid and concerted changes in the nucleo-cytoplasmic distribution of YAP.
- J. Matthew Franklin
- , Rajarshi P. Ghosh
- & Jan T. Liphardt
-
Article
| Open Access The SecA motor generates mechanical force during protein translocation
The SecA motor generates mechanical force during protein translocation
The ATPase SecA drives Sec-dependent protein translocation across the bacterial plasma membrane. Here, the authors combine kinetic translocation measurements with single-molecule force spectroscopy and demonstrate that the SecA motor generates mechanical force to unfold and translocate preproteins.
- Riti Gupta
- , Dmitri Toptygin
- & Christian M. Kaiser
-
Article
| Open Access Peroxisomal targeting of a protein phosphatase type 2C via mitochondrial transit
Peroxisomal targeting of a protein phosphatase type 2C via mitochondrial transit
Import of proteins into specific cellular compartments is critical for organelle function and several proteins are known to be imported into multiple compartments. Here, the authors report that the protein Ptc5 is first sorted to and processed in the mitochondria before being targeted to peroxisomes, which may influence mitochondria-peroxisome interorganellar contact.
- Thorsten Stehlik
- , Marco Kremp
- & Johannes Freitag
-
Article
| Open Access Nucleoplasmic signals promote directed transmembrane protein import simultaneously via multiple channels of nuclear pores
Nucleoplasmic signals promote directed transmembrane protein import simultaneously via multiple channels of nuclear pores
The contribution of central and peripheral channels of nuclear pores to transport of transmembrane proteins is unclear. Here the authors show that most inner nuclear membrane proteins use only peripheral channels, but some extend nuclear localization signals into the central channel for directed nuclear transport.
- Krishna C. Mudumbi
- , Rafal Czapiewski
- & Weidong Yang
-
Article
| Open Access Structure of the substrate-engaged SecA-SecY protein translocation machine
Structure of the substrate-engaged SecA-SecY protein translocation machine
Proteins are translocated across membranes through the Sec61/SecY channel. Here, the authors present the structure of a translocating peptide chain trapped inside the SecA-SecY complex which suggests how peptides are actively moved through the channel.
- Chengying Ma
- , Xiaofei Wu
- & Long Li
-
Article
| Open Access Structural insights into the function of type VI secretion system TssA subunits
Structural insights into the function of type VI secretion system TssA subunits
TssA is an important component of the bacterial type VI secretion system (T6SS). Here, Dix et al. integrate structural, phylogenetic and functional analysis of the TssA subunits, providing new insights into their role in T6SS assembly and function.
- Samuel R. Dix
- , Hayley J. Owen
- & Mark S. Thomas
-
Article
| Open Access Proteomics reveals signal peptide features determining the client specificity in human TRAP-dependent ER protein import
Proteomics reveals signal peptide features determining the client specificity in human TRAP-dependent ER protein import
While Sec61 enables ER import of all polypeptides with N-terminal signal peptides, only selected clients are accepted for TRAP-assisted ER import. Here, the authors use a proteomics approach to characterize TRAP-dependent clients, identifying signal peptide features that govern recognition by TRAP.
- Duy Nguyen
- , Regine Stutz
- & Richard Zimmermann
-
Article
| Open Access Revealing the mechanisms of membrane protein export by virulence-associated bacterial secretion systems
Revealing the mechanisms of membrane protein export by virulence-associated bacterial secretion systems
Many bacteria export effector proteins even when two incompatible signal sequences are present, one which would lead to export and the other to inner membrane targeting. Here the authors show that such proteins feature decreased hydrophobicity or cognate chaperone binding to prevent erroneous targeting.
- Lea Krampen
- , Silke Malmsheimer
- & Samuel Wagner
-
Article
| Open Access SUMOylation of VEGFR2 regulates its intracellular trafficking and pathological angiogenesis
SUMOylation of VEGFR2 regulates its intracellular trafficking and pathological angiogenesis
VEGFR2 is a central regulator of angiogenesis. Here Zhou et al. report that SUMOylation of VEGFR2 regulates its subcellular localisation and activity, and that endothelial-specific knockout of the SUMO endopeptidase SENP1 protects against VEGFR2-mediated pathological angiogenesis.
- Huanjiao Jenny Zhou
- , Zhe Xu
- & Wang Min
-
Article
| Open Access SKP2- and OTUD1-regulated non-proteolytic ubiquitination of YAP promotes YAP nuclear localization and activity
SKP2- and OTUD1-regulated non-proteolytic ubiquitination of YAP promotes YAP nuclear localization and activity
Regulation of Yes-associated protein (YAP) through the Hippo pathway is well established, but its Hippo-independent regulation remains to be elucidated. Here, the authors show that non-proteolytic ubiquitination presents another means of YAP regulation, promoting its nuclear localization and activity.
- Fan Yao
- , Zhicheng Zhou
- & Li Ma
-
Article
| Open Access Cyclophilin A enables specific HIV-1 Tat palmitoylation and accumulation in uninfected cells
Cyclophilin A enables specific HIV-1 Tat palmitoylation and accumulation in uninfected cells
It is not clear whether and how incoming HIV-1 Tat accumulates in uninfected cells. Here, Chopard et al. show that, in uninfected cells, incoming Tat is palmitoylated on Cys31 by DHHC-20, which increases its affinity for PI(4,5)P2 and results in its accumulation at the plasma membrane.
- Christophe Chopard
- , Phuoc Bao Viet Tong
- & Bruno Beaumelle
-
Article
| Open Access Folding of a bacterial integral outer membrane protein is initiated in the periplasm
Folding of a bacterial integral outer membrane protein is initiated in the periplasm
The Bam complex promotes the insertion of β-barrel proteins (such as UpaG, a trimeric autotransporter adhesin) into the bacterial outer membrane. Here, Sikdar et al. show that UpaG β-barrel segments fold into a trimeric structure in the periplasm before they interact with the Bam complex.
- Rakesh Sikdar
- , Janine H. Peterson
- & Harris D. Bernstein
-
Article
| Open Access Exploiting the kinesin-1 molecular motor to generate a virus membrane penetration site
Exploiting the kinesin-1 molecular motor to generate a virus membrane penetration site
How non-enveloped viruses cross host membranes is incompletely understood. Here, Ravindranet al. show that polyomavirus SV40 recruits kinesin-1 to construct a penetration site on the ER membrane.
- Madhu Sudhan Ravindran
- , Martin F. Engelke
- & Billy Tsai
-
Article
| Open Access Allosteric modulation of peroxisomal membrane protein recognition by farnesylation of the peroxisomal import receptor PEX19
Allosteric modulation of peroxisomal membrane protein recognition by farnesylation of the peroxisomal import receptor PEX19
PEX19 is a chaperone and import receptor for peroxisomal membrane proteins (PMPs). Here the authors present the structure of the farnesylated C-terminal domain of PEX19, and its interaction with PMPs reveals how the farnesyl moiety allosterically reshapes the PMP binding surface and modulates PEX19 function.
- Leonidas Emmanouilidis
- , Ulrike Schütz
- & Michael Sattler
-
Article
| Open Access Dissecting the molecular organization of the translocon-associated protein complex
Dissecting the molecular organization of the translocon-associated protein complex
The translocon-associated protein complex (TRAP) is a crucial component of the endoplasmic reticulum protein translocon. Here the authors study native translocon structures from human disease patients and algae cells to determine the molecular organization of the TRAP complex.
- Stefan Pfeffer
- , Johanna Dudek
- & Friedrich Förster
-
Article
| Open Access The non-canonical mitochondrial inner membrane presequence translocase of trypanosomatids contains two essential rhomboid-like proteins
The non-canonical mitochondrial inner membrane presequence translocase of trypanosomatids contains two essential rhomboid-like proteins
The mitochondrial protein import machinery is crucial for eukaryotes but little is known about its evolutionary origin. Here, the authors characterize the translocase of the inner membrane (TIM) in trypanosomes, showing that it contains two rhomboid-like proteins essential for protein import.
- Anke Harsman
- , Silke Oeljeklaus
- & André Schneider
-
Article
| Open Access Wnt5a induces renal AQP2 expression by activating calcineurin signalling pathway
Wnt5a induces renal AQP2 expression by activating calcineurin signalling pathway
The water channel AQP2 mediates the concentration of urine in the kidney. Here Ando et al. show that Wnt5 promotes collecting duct permeability by regulating AQP2 expression and localization through activation of the calmodulin/calcineurin signalling pathway.
- Fumiaki Ando
- , Eisei Sohara
- & Shinichi Uchida
-
Article
| Open Access Sec16 alternative splicing dynamically controls COPII transport efficiency
Sec16 alternative splicing dynamically controls COPII transport efficiency
The transport of secretory proteins from the endoplasmic reticulum to the Golgi depends on COPII-coated vesicles. Here, the authors show that activation-induced alternative splicing of Sec16 controls adaptation of COPII transport to increased secretory cargo upon T cell activation.
- Ilka Wilhelmi
- , Regina Kanski
- & Florian Heyd
-
Article
| Open Access MCOLN1 is a ROS sensor in lysosomes that regulates autophagy
MCOLN1 is a ROS sensor in lysosomes that regulates autophagy
Reactive oxygen species (ROS) damage cell components, necessitating their clearance through autophagy. Here, the authors show that ROS can induce autophagy by triggering TRPML1 to release Ca2+from the lysosomal lumen, in turn activating the autophagy and lysosomal biogenesis regulator TFEB.
- Xiaoli Zhang
- , Xiping Cheng
- & Haoxing Xu
-
Article
| Open Access Real-time quantification of protein expression at the single-cell level via dynamic protein synthesis translocation reporters
Real-time quantification of protein expression at the single-cell level via dynamic protein synthesis translocation reporters
Single cells can display large heterogeneity in gene induction. Here, Aymoz et al. present an expression reporter based on protein translocation that can accurately measure both the levels and dynamics of protein synthesis in live single cells with a temporal resolution of less than one minute.
- Delphine Aymoz
- , Victoria Wosika
- & Serge Pelet
-
Article
| Open Access Conversion of graded phosphorylation into switch-like nuclear translocation via autoregulatory mechanisms in ERK signalling
Conversion of graded phosphorylation into switch-like nuclear translocation via autoregulatory mechanisms in ERK signalling
While ERK signalling can produce switch-like cell behaviour, phosphorylation of ERK increases linearly with extracellular signals. Here, the authors solve this seeming contradiction by showing that nuclear translocation of ERK behaves in a switch-like manner and is controlled by ERK activity.
- Yuki Shindo
- , Kazunari Iwamoto
- & Koichi Takahashi
-
Article
| Open Access Leukotriene C4 is the major trigger of stress-induced oxidative DNA damage
Leukotriene C4 is the major trigger of stress-induced oxidative DNA damage
Chemotherapeutic agents elicit ER and oxidative stress as part of their mode of action. Here the authors show that chemotherapy and ER stress trigger MGST2-based biosynthesis of LTC4, whose inhibition abolishes chemotherapy- and ER stress-triggered oxidative stress and DNA damage, resulting in the attenuation of cell death.
- Efrat Dvash
- , Michal Har-Tal
- & Menachem Rubinstein
-
Article
| Open Access Mammalian SRP receptor switches the Sec61 translocase from Sec62 to SRP-dependent translocation
Mammalian SRP receptor switches the Sec61 translocase from Sec62 to SRP-dependent translocation
Sec62 is a membrane-bound protein that is involved in the translocation of proteins via the signal recognition particle-independent pathway. Here, the authors show that the receptor SRα displaces Sec62 from the translocon and isolate the domain on SRα that is responsible for this.
- Bhalchandra Jadhav
- , Michael McKenna
- & Martin R. Pool
-
Article
| Open Access Structure of the native Sec61 protein-conducting channel
Structure of the native Sec61 protein-conducting channel
The protein-conducting channel Sec61 is responsible for protein transport and membrane insertion at the endoplasmic reticulum. Here, the authors determine the structure of ribosome-bound Sec61 in a native context, in which it adopts a laterally open conformation, irrespective of its functional state.
- Stefan Pfeffer
- , Laura Burbaum
- & Friedrich Förster
-
Article
| Open Access The intellectual disability protein RAB39B selectively regulates GluA2 trafficking to determine synaptic AMPAR composition
The intellectual disability protein RAB39B selectively regulates GluA2 trafficking to determine synaptic AMPAR composition
Mutations in the RAB39Bgene, which encodes a protein involved in vesicular trafficking, are associated with intellectual disability, but the impact of RAB39B loss of function on synaptic activity is not known. Here the authors show that RAB39B interacts with PICK1, and that this interaction is critical for the translocation of AMPA receptor subunits into the Golgi.
- Maria Lidia Mignogna
- , Maila Giannandrea
- & Patrizia D’Adamo
-
Article |
 Versatile in vitro system to study translocation and functional integration of bacterial outer membrane proteins
Versatile in vitro system to study translocation and functional integration of bacterial outer membrane proteins
The mechanisms of protein translocation across and integration into bacterial outer membranes are poorly understood. Here, Norell et al. reconstitute type-V secretion and β-barrel protein biogenesis in proteoliposomes providing a versatile cell-free system to study integration and translocation.
- Derrick Norell
- , Alexander Heuck
- & Enguo Fan
-
Article |
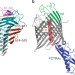 Translocation path of a substrate protein through its Omp85 transporter
Translocation path of a substrate protein through its Omp85 transporter
The two-partner secretion system transports proteins across the bacterial outer membrane but the molecular mechanisms involved are poorly understood. Here, Baud et al. use site-specific crosslinking to track the path of a protein substrate through the β-barrel of its Omp85 transporter.
- Catherine Baud
- , Jérémy Guérin
- & Françoise Jacob-Dubuisson

 Modulation of peroxisomal import by the PEX13 SH3 domain and a proximal FxxxF binding motif
Modulation of peroxisomal import by the PEX13 SH3 domain and a proximal FxxxF binding motif