Article
|
Open Access
Featured
-
-
Article
| Open Access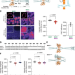 The structural flexibility of MAD1 facilitates the assembly of the Mitotic Checkpoint Complex
The structural flexibility of MAD1 facilitates the assembly of the Mitotic Checkpoint Complex
The formation of the mitotic checkpoint complex requires an intricate spatial coordination amongst the proteins Bub1, Mad1, Cdc20, and Mad2. Chen et al show that a structural flexibility in Mad1 plays an essential role in achieving this coordination.
- Chu Chen
- , Valentina Piano
- & Ajit P. Joglekar
-
Article
| Open Access Phase separation modulates the assembly and dynamics of a polarity-related scaffold-signaling hub
Phase separation modulates the assembly and dynamics of a polarity-related scaffold-signaling hub
The polarization of distinct scaffold-signaling hubs at opposite cell poles constitutes the basis of asymmetric cell division. Here, the authors show that phase separation serves as a general mechanism to regulate the assembly and dynamics of a new-pole scaffold-signaling hub.
- Wei Tan
- , Sihua Cheng
- & Wei Zhao
-
Article
| Open Access Juxtaposition of Bub1 and Cdc20 on phosphorylated Mad1 during catalytic mitotic checkpoint complex assembly
Juxtaposition of Bub1 and Cdc20 on phosphorylated Mad1 during catalytic mitotic checkpoint complex assembly
Formation of the mitotic checkpoint complex (MCC) is catalysed by a phosphorylation-dependent scaffold. This work provides structural details of how a tripartite Mad1:Bub1:Cdc20 complex presents Cdc20 to Mad2, triggering open-to-closed conversion of Mad2 to assemble the MCC.
- Elyse S. Fischer
- , Conny W. H. Yu
- & David Barford
-
Article
| Open Access Polyanions provide selective control of APC/C interactions with the activator subunit
Polyanions provide selective control of APC/C interactions with the activator subunit
Activation of APC/C depends on transient interactions with an activator subunit. Here, the authors show that cellular polyanions trigger APC/C-activator dissociation and that high-affinity substrates block this effect, suggesting that substrate availability controls APC/C-activator binding.
- Arda Mizrak
- & David O. Morgan
-
Article
| Open Access Coupling of Polo kinase activation to nuclear localization by a bifunctional NLS is required during mitotic entry
Coupling of Polo kinase activation to nuclear localization by a bifunctional NLS is required during mitotic entry
Drosophila Polo kinase is the founding member of the Polo-Like Kinase (PLK) family and a master regulator of mitosis and cytokinesis. Here the authors uncover a molecular mechanism for the spatiotemporal regulation of Polo kinase during mitotic entry through a phosphorylation event that triggers nuclear import.
- David Kachaner
- , Damien Garrido
- & Vincent Archambault
-
Article
| Open Access Molecular requirements for the inter-subunit interaction and kinetochore recruitment of SKAP and Astrin
Molecular requirements for the inter-subunit interaction and kinetochore recruitment of SKAP and Astrin
SKAP and Astrin form a heterodimer that localizes to spindle microtubules and to mature microtubule-kinetochore attachments during mitosis. Here, the authors identify molecular requirements for the inter-subunit interaction of SKAP and Astrin and kinetochore recruitment.
- Alexandra Friese
- , Alex C. Faesen
- & Andrea Musacchio
-
Article
| Open Access Cdk1 activity acts as a quantitative platform for coordinating cell cycle progression with periodic transcription
Cdk1 activity acts as a quantitative platform for coordinating cell cycle progression with periodic transcription
Cell proliferation is regulated by cyclin-dependent kinases (Cdks) and relies on periodic gene cluster expression according to cell cycle phases. Here the authors use a synthetic regulatable Cdk1 module to demonstrate that periodic expression is governed by quantitative changes in Cdk1 activity.
- Gabor Banyai
- , Feriel Baïdi
- & Zsolt Szilagyi
-
Article
| Open Access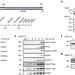 Interphase APC/C–Cdc20 inhibition by cyclin A2–Cdk2 ensures efficient mitotic entry
Interphase APC/C–Cdc20 inhibition by cyclin A2–Cdk2 ensures efficient mitotic entry
The Anaphase Promoting Complex/Cyclosome (APC/C) and its co-activator Cdc20 regulate mitotic progression, but both are also present in interphase. Here Hein and Nilsson show that Cyclin A2–CDK2 phosphorylates Cdc20 to inhibit APC/C–Cdc20 activity during this cell cycle phase to promote mitotic entry.
- Jamin B. Hein
- & Jakob Nilsson
-
Article
| Open Access Cdk1 orders mitotic events through coordination of a chromosome-associated phosphatase switch
Cdk1 orders mitotic events through coordination of a chromosome-associated phosphatase switch
RepoMan is a signalling scaffold for mitotic phosphatases PP1 and PP2A-B56, which regulate targeting of Aurora B and RepoMan respectively, to the chromosomes. Here Qian et al.show that Cdk1 phosphorylates RepoMan to modulate the binding of PP1 and PP2A-B56, contributing to orderly mitotic progression.
- Junbin Qian
- , Monique Beullens
- & Mathieu Bollen
-
Article
| Open Access PICH promotes sister chromatid disjunction and co-operates with topoisomerase II in mitosis
PICH promotes sister chromatid disjunction and co-operates with topoisomerase II in mitosis
During mitosis the translocase PICH binds to ultrafine bridges formed from DNA catenanes that are unresolved by topoisomerase II. In this study, the authors show that PICH stimulates toposiomerase II activity and that they cooperate to resolve these structures.
- Christian F. Nielsen
- , Diana Huttner
- & Ian D. Hickson
-
Article
| Open Access CDK1 structures reveal conserved and unique features of the essential cell cycle CDK
CDK1 structures reveal conserved and unique features of the essential cell cycle CDK
Cyclin-dependent kinases are the principal drivers of cell cycle progression. Here the authors present several crystal structures of Cdk1 in complex with cyclin B and/or the assembly factors Cks1/2 and a small molecule inhibitor to reveal key features of this essential mitotic kinase.
- Nicholas R. Brown
- , Svitlana Korolchuk
- & Jane A. Endicott
-
Article
| Open Access Division of labour between Myc and G1 cyclins in cell cycle commitment and pace control
Division of labour between Myc and G1 cyclins in cell cycle commitment and pace control
The transcription factor E2F is critical for determining cell proliferation. By monitoring E2F activity in single cells throughout the cell cycle, Dong et al.provide evidence that Myc and G1 cyclin/CDKs regulate different aspects of E2F temporal dynamics, resulting in distinct phenotypic outputs.
- Peng Dong
- , Manoj V. Maddali
- & Lingchong You
-
Article |
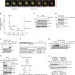 Pre-anaphase chromosome oscillations are regulated by the antagonistic activities of Cdk1 and PP1 on Kif18A
Pre-anaphase chromosome oscillations are regulated by the antagonistic activities of Cdk1 and PP1 on Kif18A
As chromosomes align during metaphase, they initially oscillate back and forth before settling at the metaphase plate. Häfner et al. show that antagonistic phosphorylation and dephosphorylation of the kinesin Kif18A underlies this behaviour and is required for accurate sister chromatid segregation.
- Julia Häfner
- , Monika I. Mayr
- & Thomas U. Mayer
-
Article
| Open Access Greatwall kinase and cyclin B-Cdk1 are both critical constituents of M-phase-promoting factor
Greatwall kinase and cyclin B-Cdk1 are both critical constituents of M-phase-promoting factor
Cyclin B–Cdk1 is thought to be synonymous with the promoting factor that drives entry into M-phase of the cell cycle. Here, Greatwall kinase is shown to be required for the breakdown of the nuclear envelope and the assembly of the spindle on entry into M-phase, suggesting that it too is a part of the M-phase-promoting factor.
- Masatoshi Hara
- , Yusuke Abe
- & Takeo Kishimoto
-
Article |
 Cyclin B-dependent kinase 1 regulates human TRF1 to modulate the resolution of sister telomeres
Cyclin B-dependent kinase 1 regulates human TRF1 to modulate the resolution of sister telomeres
TRF1 is a telomere binding protein involved in sister telomere cohesion. In this study, the ability of TRF1 to bind to telomeres in mitosis is inhibited by cyclin-dependent kinase 1-mediated phosphorylation, which may facilitate sister telomere resolution during mitosis.
- Megan McKerlie
- & Xu-Dong Zhu
-
Article
| Open Access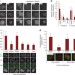 Aurora B potentiates Mps1 activation to ensure rapid checkpoint establishment at the onset of mitosis
Aurora B potentiates Mps1 activation to ensure rapid checkpoint establishment at the onset of mitosis
Mitotic exit is controlled by a cell division checkpoint that prevents premature degradation of cyclin B by the anaphase-promoting complex. Saurinet al. show that Aurora B directly regulates timely establishment of this checkpoint by facilitating activation of Mps1 kinase at unattached kinetochores.
- Adrian T. Saurin
- , Maike S. van der Waal
- & Geert J.P.L. Kops

 An interphase actin wave promotes mitochondrial content mixing and organelle homeostasis
An interphase actin wave promotes mitochondrial content mixing and organelle homeostasis