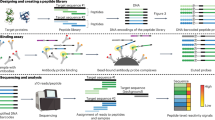
Abstract
We describe general methods to detect and quantify small numbers of specific molecules. We redirected self-splicing protein inteins to create 'tadpoles', chimeric molecules comprised of a protein head covalently coupled to an oligonucleotide tail. We made different classes of tadpoles that bind specific targets, including Bacillus anthracis protective antigen and the enzyme cofactor biotin. We measured the amount of bound target by quantifying DNA tails by T7 RNA polymerase runoff transcription and real-time polymerase chain reaction (PCR) evaluated by rigorous statistical methods. These assays had a dynamic range of detection of more than 11 orders of magnitude and distinguished numbers of molecules that differed by as little as 10%. At their low limit, these assays were used to detect as few as 6,400 protective antigen molecules, 600 biotin molecules and 150 biotinylated protein molecules. In crudely fractionated human serum, the assays were used to detect as few as 32,000 protective antigen molecules. Tadpoles thus enable sensitive detection and precise quantification of molecules other than DNA and RNA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others

References
Engvall, E. & Perlman, P. Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 8, 871–874 (1971).
Yalow, R.S. & Berson, S.A. Assay of plasma insulin in human subjects by immunological methods. Nature 184 Suppl. 21, 1648–1649 (1959).
Brenner, S. & Lerner, R.A. Encoded combinatorial chemistry. Proc. Natl. Acad. Sci. USA 89, 5381–5383 (1992).
Sano, T., Smith, C.L. & Cantor, C.R. Immuno-PCR: very sensitive antigen detection by means of specific antibody-DNA conjugates. Science 258, 120–122 (1992).
Fredriksson, S. et al. Protein detection using proximity-dependent DNA ligation assays. Nat. Biotechnol. 20, 473–477 (2002).
Hendrickson, E.R., Truby, T.M., Joerger, R.D., Majarian, W.R. & Ebersole, R.C. High sensitivity multianalyte immunoassay using covalent DNA-labeled antibodies and polymerase chain reaction. Nucleic Acids Res. 23, 522–529 (1995).
McKie, A., Samuel, D., Cohen, B. & Saunders, N.A. A quantitative immuno-PCR assay for the detection of mumps-specific IgG. J. Immunol. Methods 270, 135–141 (2002).
Niemeyer, C.M. et al. Self-assembly of DNA-streptavidin nanostructures and their use as reagents in immuno-PCR. Nucleic Acids Res. 27, 4553–4561 (1999).
Zhang, H.T., Kacharmina, J.E., Miyashiro, K., Greene, M.I. & Eberwine, J. Protein quantification from complex protein mixtures using a proteomics methodology with single-cell resolution. Proc. Natl. Acad. Sci. USA 98, 5497–5502 (2001).
Nam, J.M., Thaxton, C.S. & Mirkin, C.A. Nanoparticle-based bio-bar codes for the ultrasensitive detection of proteins. Science 301, 1884–1886 (2003).
Perler, F.B. et al. Intervening sequences in an Archaea DNA polymerase gene. Proc. Natl. Acad. Sci. USA 89, 5577–5581 (1992).
Muir, T., Sondhi, D. & Cole, P. Expressed protein ligation: A general method for protein engineering. Proc. Natl. Acad. Sci. USA 95, 6705–6710 (1998).
Sydor, J.R., Mariano, M., Sideris, S. & Nock, S. Establishment of intein-mediated protein ligation under denaturing conditions: C-terminal labeling of a single-chain antibody for biochip screening. Bioconjug. Chem. 13, 707–712 (2002).
Lovrinovic, M. et al. Synthesis of protein-nucleic acid conjugates by expressed protein ligation. Chem. Commun. (Camb.) 7, 822–823 (2003).
Green, N.M. & Melamed, M.D. Optical rotatory dispersion, circular dichroism and far-ultraviolet spectra of avidin and streptavidin. Biochem. J. 100, 614–621 (1966).
Huston, J.S. et al. Protein engineering of antibody binding sites: recovery of specific activity in an anti-digoxin single-chain Fv analogue produced in Escherichia coli. Proc. Natl. Acad. Sci. USA 85, 5879–5883 (1988).
Colas, P. et al. Genetic selection of peptide aptamers that recognize and inhibit cyclin-dependent kinase 2. Nature 380, 548–550 (1996).
Watanabe, T. et al. The roles of the C-terminal domain and type III domains of chitinase A1 from Bacillus circulans WL-12 in chitin degradation. J. Bacteriol. 176, 4465–4472 (1994).
Stetsenko, D.A. & Gait, M.J. New phosphoramidite reagents for the synthesis of oligonucleotides containing a cysteine residue useful in peptide conjugation. Nucleosides Nucleotides Nucleic Acids 19, 1751–1764 (2000).
Chong, S. et al. Single-column purification of free recombinant proteins using a self-cleavable affinity tag derived from a protein splicing element. Gene 192, 271–281 (1997).
Sano, T., Pandori, M.W., Chen, X., Smith, C.L. & Cantor, C.R. Recombinant core streptavidins. A minimum-sized core streptavidin has enhanced structural stability and higher accessibility to biotinylated macromolecules. J. Biol. Chem. 270, 28204–28209 (1995).
Green, N.M. A Spectrophotometric Assay for Avidin and Biotin Based on Binding of Dyes by Avidin. Biochem. J. 94, 23C–24C (1965).
Van Gelder, R.N. et al. Amplified RNA synthesized from limited quantities of heterogeneous cDNA. Proc. Natl. Acad. Sci. USA 87, 1663–1667 (1990).
Higuchi, R., Fockler, C., Dollinger, G. & Watson, R. Kinetic PCR analysis: real-time monitoring of DNA amplification reactions. Biotechnology 11, 1026–1030 (1993).
Timkovich, R. Detection of the stable addition of carbodiimide to proteins. Anal. Biochem. 79, 135–143 (1977).
Staros, J.V., Wright, R.W. & Swingle, D.M. Enhancement by N-hydroxysulfosuccinimide of water-soluble carbodiimide- mediated coupling reactions. Anal. Biochem. 156, 220–222 (1986).
Neyman, J. Outline of a theory of statistical estimation based on the classical theory of probability. Philos. Trans. R. Soc. London A236, 333–380 (1937).
Feldman, G.J. & Cousins, R.D. Unified approach to the classical statistical analysis of small signals. Phys. Rev. 57, 3873–3889 (1998).
Fabbrizio, E. et al. Inhibition of mammalian cell proliferation by genetically selected peptide aptamers that functionally antagonize E2F activity. Oncogene 18, 4357–4363 (1999).
Leppla, S.H. Anthrax toxin edema factor: a bacterial adenylate cyclase that increases cyclic AMP concentrations of eukaryotic cells. Proc. Natl. Acad. Sci. USA 79, 3162–3166 (1982).
Maynard, J.A. et al. Protection against anthrax toxin by recombinant antibody fragments correlates with antigen affinity. Nat. Biotechnol. 20, 597–601 (2002).
Prinz, W.A., Aslund, F., Holmgren, A. & Beckwith, J. The role of the thioredoxin and glutaredoxin pathways in reducing protein disulfide bonds in the Escherichia coli cytoplasm. J. Biol. Chem. 272, 15661–15667 (1997).
Salmon, S.E., Mackey, G. & Fudenberg, H.H. Sandwich” solid phase radioimmunoassay for the quantitative deterimination of human immunoglobulins. J. Immunol. 103, 129–137 (1969).
Chen, L., Benner, J. & Perler, F.B. Protein splicing in the absence of an intein penultimate histidine. J. Biol. Chem. 275, 20431–20435 (2000).
Schafer, U., Beck, K. & Muller, M. Skp, a molecular chaperone of gram-negative bacteria, is required for the formation of soluble periplasmic intermediates of outer membrane proteins. J. Biol. Chem. 274, 24567–24574 (1999).
Hayhurst, A. & Harris, W.J. Escherichia coli skp chaperone coexpression improves solubility and phage display of single-chain antibody fragments. Protein Expr. Purif. 15, 336–343 (1999).
Bothmann, H. & Pluckthun, A. Selection for a periplasmic factor improving phage display and functional periplasmic expression. Nat. Biotechnol. 16, 376–380 (1998).
Caruthers, M.H. et al. New chemical methods for synthesizing polynucleotides. Nucleic Acids Symp. Ser. 7, 215–223 (1980).
Caruthers, M.H. et al. Deoxyoligonucleotide synthesis via the phosphoramidite method. Gene Amplif. Anal. 3, 1–26 (1983).
Tuma, R.S. et al. Characterization of SYBR Gold nucleic acid gel stain: a dye optimized for use with 300-nm ultraviolet transilluminators. Anal. Biochem. 268, 278–288 (1999).
Zacharieva, E.I., Georgieva, M.P., Kabaivanov, V.S. & Popov, D.V. Preparation and investigation of cross-linked copolymers containing antitumour agents. Biomaterials 4, 197–200 (1983).
Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Garner, D.L., Johnson, L.A., Yue, S.T., Roth, B.L. & Haugland, R.P. Dual DNA staining assessment of bovine sperm viability using SYBR-14 and propidium iodide. J. Androl. 15, 620–629 (1994).
Acknowledgements
The authors thank R. Fu for technical assistance with early real-time PCR experiments, L. Lok for invaluable advice on statistical analysis, O. Resnekov for suggesting T-gel for fractionating serum, C. Cantor for the gene encoding the streptavidin core subunit, J. Maynard for plasmids encoding anti-PA scFvs, purified scFvs and antibodies, and A. Coleman-Lerner, K. Benjamin, A. Gordon, L. Lok, A. Mendelsohn, O. Resnekov, E. Serra and R. Yu for useful discussions and comments on the manuscript. Some support for this work came from grants from the National Science Foundation and Defense Advanced Research Projects Agency, but main support came from the Center for Genomic Experimentation and Computation, a National Institutes of Health Center of Excellence in Genomic Science (P50 HG02370).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The technology described in this manuscript has pending patents and may be licensed for use.
Supplementary information
Supplementary Fig. 1
Verification of gaussian distribution. (PDF 176 kb)
Supplementary Fig. 2
Biotin enyzyme linked immuno-adsorption assay. (PDF 57 kb)
Supplementary Fig. 3
PA enyzyme linked immuno-adsorption assay. (PDF 61 kb)
Supplementary Table 1
Affinity protein heads. (PDF 66 kb)
Supplementary Table 2
DNA tails used to construct tadpoles. (PDF 51 kb)
Supplementary Table 3
Forward and reverse PCR primer sequences used to amplify specific tadpole tail DNAs. (PDF 50 kb)
Supplementary Table 4
Evanescent wave measurements of tadpole binding. (PDF 136 kb)
Rights and permissions
About this article
Cite this article
Burbulis, I., Yamaguchi, K., Gordon, A. et al. Using protein-DNA chimeras to detect and count small numbers of molecules. Nat Methods 2, 31–37 (2005). https://doi.org/10.1038/nmeth729
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth729
This article is cited by
-
Advances in Anthrax Detection: Overview of Bioprobes and Biosensors
Applied Biochemistry and Biotechnology (2015)
-
Ratiometric electrochemical proximity assay for sensitive one-step protein detection
Scientific Reports (2014)
-
Sensitive monitoring of RNA transcription levels using a graphene oxide fluorescence switch
Chinese Science Bulletin (2013)
-
Artificial Protein Complexes for Biocatalysis
Topics in Catalysis (2012)
-
Quantifying small numbers of antibodies with a 'near-universal' protein-DNA chimera
Nature Methods (2007)