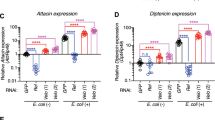
Abstract
IκB kinase (IKK) and Jun N-terminal kinase (Jnk) signaling modules are important in the synthesis of immune effector molecules during innate immune responses against lipopolysaccharide and peptidoglycan. However, the regulatory mechanisms required for specificity and termination of these immune responses are unclear. We show here that crosstalk occurred between the drosophila Jnk and IKK pathways, which led to downregulation of each other's activity. The inhibitory action of Jnk was mediated by binding of drosophila activator protein 1 (AP1) to promoters activated by the transcription factor NF-κB. This binding led to recruitment of the histone deacetylase dHDAC1 to the promoter of the gene encoding the antibacterial protein Attacin-A and to local modification of histone acetylation content. Thus, AP1 acts as a repressor by recruiting the deacetylase complex to terminate activation of a group of NF-κB target genes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Kimbrell, D.A. & Beutler, B. The evolution and genetics of innate immunity. Nat. Rev. Genet. 2, 256–267 (2001).
Hoffmann, J.A. The immune response of Drosophila. Nature 426, 33–38 (2003).
Tzou, P., De Gregorio, E. & Lemaitre, B. How Drosophila combats microbial infection: a model to study innate immunity and host-pathogen interactions. Curr. Opin. Microbiol. 5, 102–110 (2002).
Medzhitov, R. & Janeway, C.A., Jr. Innate immunity. N. Engl. J. Med. 343, 338–344 (2000).
Janeway, C.A., Jr. & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197–216 (2002).
Brennan, C.A. & Anderson, K.V. Drosophila: the genetics of innate immune recognition and response. Annu. Rev. Immunol. 22, 457–483 (2004).
Stronach, B.E. & Perrimon, N. Stress signaling in Drosophila. Oncogene 18, 6172–6182 (1999).
Boutros, M., Agaisse, H. & Perrimon, N. Sequential activation of signaling pathways during innate immune responses in Drosophila. Dev. Cell 3, 711–722 (2002).
Hoffmann, J.A. & Reichhart, J.M. Drosophila innate immunity: an evolutionary perspective. Nat. Immunol. 3, 121–126 (2002).
Hultmark, D. Drosophila immunity: paths and patterns. Curr. Opin. Immunol. 15, 12–19 (2003).
Wasserman, S.A. A conserved signal transduction pathway regulating the activity of the rel-like proteins dorsal and NF-κB. Mol. Biol. Cell 4, 767–771 (1993).
Lemaitre, B., Nicolas, E., Michaut, L., Reichhart, J.M. & Hoffmann, J.A. The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell 86, 973–983 (1996).
Meng, X., Khanuja, B.S. & Ip, Y.T. Toll receptor-mediated Drosophila immune response requires Dif, an NF-κB factor. Genes Dev. 13, 792–797 (1999).
Wasserman, S.A. Toll signaling: the enigma variations. Curr. Opin. Genet. Dev. 10, 497–502 (2000).
Levashina, E.A. et al. Constitutive activation of toll-mediated antifungal defense in serpin-deficient Drosophila. Science 285, 1917–1919 (1999).
Weber, A.N. et al. Binding of the Drosophila cytokine Spatzle to Toll is direct and establishes signaling. Nat. Immunol. 4, 794–800 (2003).
Ip, Y.T. et al. Dif, a dorsal-related gene that mediates an immune response in Drosophila. Cell 75, 753–763 (1993).
Tauszig-Delamasure, S., Bilak, H., Capovilla, M., Hoffmann, J.A. & Imler, J.L. Drosophila MyD88 is required for the response to fungal and Gram-positive bacterial infections. Nat. Immunol. 3, 91–97 (2002).
Nicolas, E., Reichhart, J.M., Hoffmann, J.A. & Lemaitre, B. In vivo regulation of the IκB homologue cactus during the immune response of Drosophila. J. Biol. Chem. 273, 10463–10469 (1998).
Imler, J.L. & Hoffmann, J.A. Toll receptors in Drosophila: a family of molecules regulating development and immunity. Curr. Top. Microbiol. Immunol. 270, 63–79 (2002).
Kaneko, T. et al. Monomeric and polymeric gram-negative peptidoglycan but not purified LPS stimulate the Drosophila Imd pathway. Immunity 20, 637–649 (2004).
Leulier, F. et al. The Drosophila immune system detects bacteria through specific peptidoglycan recognition. Nat. Immunol. 4, 478–484 (2003).
De Gregorio, E., Spellman, P.T., Tzou, P., Rubin, G.M. & Lemaitre, B. The Toll and Imd pathways are the major regulators of the immune response in Drosophila. EMBO J. 21, 2568–2579 (2002).
Georgel, P. et al. Drosophila immune deficiency (Imd) is a death domain protein that activates antibacterial defense and can promote apoptosis. Dev. Cell 1, 503–514 (2001).
Onfelt Tingvall, T., Roos, E. & Engstrom, Y. The imd gene is required for local Cecropin expression in Drosophila barrier epithelia. EMBO Rep. 2, 239–243 (2001).
Lemaitre, B. et al. A recessive mutation, immune deficiency (imd), defines two distinct control pathways in the Drosophila host defense. Proc. Natl. Acad. Sci. USA 92, 9465–9469 (1995).
Choe, K.M., Werner, T., Stoven, S., Hultmark, D. & Anderson, K.V. Requirement for a peptidoglycan recognition protein (PGRP) in Relish activation and antibacterial immune responses in Drosophila. Science 296, 359–362 (2002).
Gottar, M. et al. The Drosophila immune response against Gram-negative bacteria is mediated by a peptidoglycan recognition protein. Nature 416, 640–644 (2002).
Vidal, S. et al. Mutations in the Drosophila dTAK1 gene reveal a conserved function for MAPKKKs in the control of rel/NF-κB-dependent innate immune responses. Genes Dev. 15, 1900–1912 (2001).
Rutschmann, S. et al. Role of Drosophila IKKγ in a Toll-independent antibacterial immune response. Nat. Immunol. 1, 342–347 (2000).
Lu, Y., Wu, L.P. & Anderson, K.V. The antibacterial arm of the Drosophila innate immune response requires an IκB kinase. Genes Dev. 15, 104–110 (2001).
Silverman, N. et al. A Drosophila IκB kinase complex required for Relish cleavage and antibacterial immunity. Genes Dev. 14, 2461–2471 (2000).
Stoven, S. et al. Caspase-mediated processing of the Drosophila NF-κB factor Relish. Proc. Natl. Acad. Sci. USA 100, 5991–5996 (2003).
Silverman, N. et al. Immune activation of NF-κB and JNK requires Drosophila TAK1. J. Biol. Chem. 278, 48928–48934 (2003).
Stronach, B. & Perrimon, N. Activation of the JNK pathway during dorsal closure in Drosophila requires the mixed lineage kinase, slipper. Genes Dev. 16, 377–387 (2002).
Goberdhan, D.C. & Wilson, C. JNK, cytoskeletal regulator and stress response kinase? A Drosophila perspective. Bioessays 20, 1009–1019 (1998).
Tang, G. et al. Inhibition of JNK activation through NF-κB target genes. Nature 414, 313–317 (2001).
De Smaele, E. et al. Induction of gadd45β by NF-κB downregulates pro-apoptotic JNK signalling. Nature 414, 308–313 (2001).
Park, J.M. et al. Targeting of TAK1 by the NF-κB protein Relish regulates the JNK-mediated immune response in Drosophila. Genes Dev. 18, 584–594 (2004).
Boutros, M. et al. Genome-wide RNAi analysis of growth and viability in Drosophila cells. Science 303, 832–835 (2004).
Ciapponi, L. & Bohmann, D. An essential function of AP-1 heterodimers in Drosophila development. Mech. Dev. 115, 35–40 (2002).
Uvell, H. & Engstrom, Y. Functional characterization of a novel promoter element required for an innate immune response in Drosophila. Mol. Cell. Biol. 23, 8272–8281 (2003).
Dushay, M.S. et al. Two attacin antibacterial genes of Drosophila melanogaster. Gene 246, 49–57 (2000).
Smith, E.R. et al. Cloning of Drosophila GCN5: conserved features among metazoan GCN5 family members. Nucleic Acids Res. 26, 2948–2954 (1998).
Huang, X. & Kadonaga, J.T. Biochemical analysis of transcriptional repression by Drosophila histone deacetylase 1. J. Biol. Chem. 276, 12497–12500 (2001).
Kim, T.W. et al. MED16 and MED23 of Mediator are coactivators of lipopolysaccharide- and heat-shock-induced transcriptional activators. Proc. Natl. Acad. Sci. USA 101, 12153–12158 (2004).
Ventura, J.J., Kennedy, N.J., Flavell, R.A. & Davis, R.J. JNK regulates autocrine expression of TGF-β1. Mol. Cell 15, 269–278 (2004).
Weiss, C. et al. JNK phosphorylation relieves HDAC3-dependent suppression of the transcriptional activity of c-Jun. EMBO J. 22, 3686–3695 (2003).
Barlev, A.N. et al. Acetylation of p53 activates transcription through recruitment of coactivators/histone acetyltransferases. Mol. Cell 8, 1243–1254 (2001).
Chen, L.-F. & Greene, C.W. Regulation of distinct biological activities of the NF-κB transcription factor complex by acetylation. J. Mol. Med. 81, 549–557 (2003).
Acknowledgements
We thank W.J. Lee (Ewha Womans University, Seoul, Korea) for providing immunocompetent SL2 cells; J.L. Imler (Institute de Biologie Moleculaire et Cellulaire, Strasbourg, France) for providing reporter plasmids; and J.M. Park (University of California at San Diego) for helpful advice. BRB-ArrayTools v3.X developed by R. Simon (National Cancer Institutes of Health, Bethesda, Maryland) and A. Peng Lam (EMMES, Rockville, Maryland) were used for microarray analyses. Supported by the Creative Research Initiatives Program of the Korean Ministry of Science and Technology (Y.-J.K.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Y.H. Song, S.N. Kim and J.H. Yoon are employed by Digital Genomics.
Supplementary information
Supplementary Fig. 1
Anti-MED20 specifically recognized drosophila MED20. (PDF 159 kb)
Supplementary Table 1
DNA chip analysis of LPS response if SL2 cells and the effect of cyclohexamide on LPS induction. (PDF 97 kb)
Supplementary Table 2
Expression profile defects of the LPS induced genes in the imd-, bsk- and Rel-RNAi treated cells. (PDF 119 kb)
Rights and permissions
About this article
Cite this article
Kim, T., Yoon, J., Cho, H. et al. Downregulation of lipopolysaccharide response in drosophila by negative crosstalk between the AP1 and NF-κB signaling modules. Nat Immunol 6, 211–218 (2005). https://doi.org/10.1038/ni1159
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1159
This article is cited by
-
Embryogenesis and apoptosis appears as key molecular pathways involved in Varroa destructor reproduction
Apidologie (2023)
-
Regulatory regions in natural transposable element insertions drive interindividual differences in response to immune challenges in Drosophila
Genome Biology (2021)
-
Inhibition of semaphorin-3a suppresses lipopolysaccharide-induced acute kidney injury
Journal of Molecular Medicine (2018)
-
A roadmap of constitutive NF-κB activity in Hodgkin lymphoma: Dominant roles of p50 and p52 revealed by genome-wide analyses
Genome Medicine (2016)
-
Proteomic Analysis of Silkworm Antennae
Journal of Chemical Ecology (2015)